For Figure 19-6, describe any changes in mass that might be detected at the Pt, Cu, and
Question:
For Figure 19-6, describe any changes in mass that might be detected at the Pt, Cu, and Zn electrodes as electric current passes through the electrochemical cells.
Figure 19-6
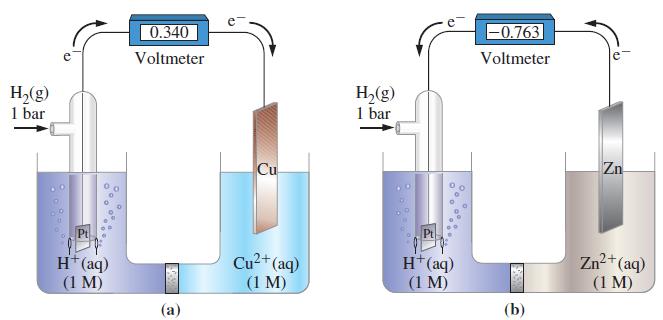
Transcribed Image Text:
H₂(g) 1 bar Pt H+ (aq) (1 M) 0.340 Voltmeter (a) Cu Cu²+ (aq) (1 M) H₂(g) 1 bar H+ (aq) (1 M) -0.763 Voltmeter (b) Zn Zn²+ (aq) (1 M)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 83% (6 reviews)
In Figure 196 we have two different electrochemical cells both containing the same electrolyte solut...View the full answer

Answered By

Asd fgh
sadasmdna,smdna,smdna,msdn,masdn,masnd,masnd,m asd.as,dmas,dma.,sd as.dmas.,dma.,s ma.,sdm.,as mda.,smd.,asmd.,asmd.,asmd.,asm
5.00+
1+ Reviews
15+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
As shown in Fig.P1.41m, air is contained in a vertical piston-cylinder assembly fitted with an electrical resistor. The atmosphere exerts a pressure of 14.lbf/in2 on the top piston, which has a mass...
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
An electric cable of radius r l and thermal conductivity k e is enclosed by an insulating sleeve whose outer surface is of radius r 2 and experiences convection heat transfer and radiation exchange...
-
Write a formula for a function g whose graph is similar to f(x) but satisfies the given conditions. Do not simplify the formula. f(x) = 3x 3x + 2 (a) Shifted right 2000 units and upward 70 units (b)...
-
Refer to CVS Corporations annual report in the Supplement to Chapter 5 to answer the following questions. 1. Consolidated balance sheets: a. Did the amount of working capital increase or decrease...
-
The operations manager for Cozine Corporation is concerned with variation in the number of pounds of garbage collected per truck. If this variation is too high, the manager will change the truck...
-
Understand why and how firms internationalize. LO.1
-
The actual cash received from cash sales was $11,279, and the amount indicated by the cash register total was $11,256. a. What is the amount deposited in the bank for the days sales? b. What is...
-
Scenario 3: Paula is a single taxpayer and loves to go to flea markets, garage sales, and antique shows. Five years ago she purchased a small oil painting at a garage sale for $20. Two months ago she...
-
(A) The cell diagram for an electrochemical cell is written as Write the equations for the half-cell reactions that occur at the electrodes. Balance the overall cell reaction. (B) The cell diagram...
-
The cell diagram for an electrochemical cell is written as Write the equations for the half-cell reactions that occur at the electrodes. Balance the overall cell reaction. Ni(s)...
-
Imagine that you and a partner are planning to launch a business that sells healthy lunch choices from an on-campus food truck. You'll need seed capital for your venture. Outline how you would...
-
(a)The local police station found that the speed of vehicles travelling around the suburb in the 60 km/hour zone varies uniformly between 55 km/hour and 62 km/hour. What is the probability that the...
-
Consider the following fixed-point iteration: xn+1 = g(xn), where [f(x)] 2 g(x) = x (x + f(x)) f(x)* (a) What is the order of convergence for the method? (e.g. what is p?). Hint: Show that the method...
-
Problem 1. In a study of infant birth weight and maternal factors, the newborn babies were categorized as being either small size for gestational age (N=201) or normal size (N=2089). The following...
-
Max 1 page allowed] Consider a DRAM chip of capacity 256 KB and each memory location contains 8 bits. The memory chip is organized in matrix form with equal number of rows and column for each memory...
-
find the dimensions of a notman window of perimeter 3 9 ft that will admit the greatest possible amount of light. Round answer to two decimal places
-
Nero Mines paid $432,000 for the right to extract ore from a 425,000-ton mineral deposit. In addition to the purchase price, Nero Mines also paid a $150 filing fee, a $2,700 license fee to the state...
-
Wal-Mart is the second largest retailer in the world. The data file on the disk holds monthly data on Wal-Marts revenue, along with several possibly related economic variables. a) Using computer...
-
DeWeese Corporation issues $400,000 of 8%, 5-year bonds on January 1, 2010, at 105. Assuming that the straight-line method is used to amortize the premium, what is the total amount of interest...
-
Mareska Inc. is considering two alternatives to finance its construction of a new $2 million plant. (a) Issuance of 200,000 shares of common stock at the market price of $10 per share. (b) Issuance...
-
Pruitt Corporation issued 3,000, 8%, 5-year, $1,000 bonds dated January 1, 2010, at 100. (a) Prepare the journal entry to record the sale of these bonds on January 1, 2010. (b) Prepare the journal...
-
You are the digital marketing director for High West fashions, a regional clothing company that specializes in custom t-shirts. Your company has decided to launch an online advertising campaign that...
-
In-the-money put options will automatically get exercised at the expiration. True OR False
-
Which of the following examples of business-use property is NOT eligible for Section 1231 treatment when sold at a gain? * Sale of land held for three years. Net gain from a casualty gain on a dump...

Study smarter with the SolutionInn App


