From the data in Table 20.2 and Figure 20-2 for the decomposition of H 2 O 2
Question:
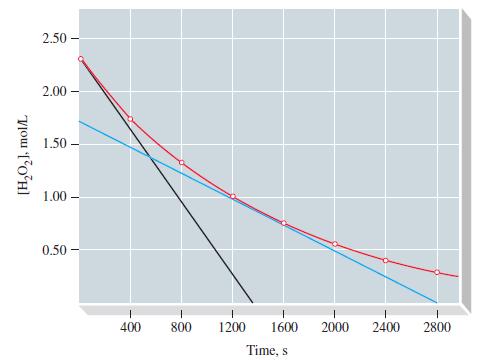
From the data in Table 20.2 and Figure 20-2 for the decomposition of H2O2, determine (a) the initial rate of reaction, and (b) [H2O2]t at t = 100 s, assuming that the initial rate is constant for at least 100 s.
Table 20.2
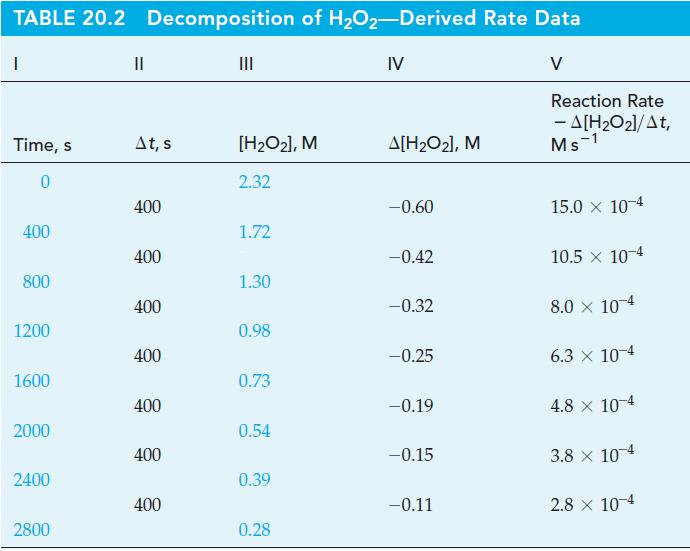
Figure 20-2
Transcribed Image Text:
TABLE 20.2 Decomposition of H₂O2-Derived Rate Data IV I Time, s 0 400 800 1200 1600 2000 2400 2800 II At, s 400 400 400 400 400 400 400 ||| [H₂O₂], M 2.32 1.72 1.30 0.98 0.73 0.54 0.39 0.28 A[H₂O₂], M -0.60 -0.42 -0.32 -0.25 -0.19 -0.15 -0.11 V Reaction Rate - A[H₂O₂]/At, Ms 1 15.0 x 10-4 10.5 x 10-4 8.0 × 10-4 6.3 x 10-4 4.8 x 10-4 3.8 x 10-4 2.8 x 10-4
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (4 reviews)
Analyze To determine the initial rate of reaction from the slope of the tangent line in part a we us...View the full answer

Answered By

Ashington Waweru
I am a lecturer, research writer and also a qualified financial analyst and accountant. I am qualified and articulate in many disciplines including English, Accounting, Finance, Quantitative spreadsheet analysis, Economics, and Statistics. I am an expert with sixteen years of experience in online industry-related work. I have a master's in business administration and a bachelor’s degree in education, accounting, and economics options.
I am a writer and proofreading expert with sixteen years of experience in online writing, proofreading, and text editing. I have vast knowledge and experience in writing techniques and styles such as APA, ASA, MLA, Chicago, Turabian, IEEE, and many others.
I am also an online blogger and research writer with sixteen years of writing and proofreading articles and reports. I have written many scripts and articles for blogs, and I also specialize in search engine
I have sixteen years of experience in Excel data entry, Excel data analysis, R-studio quantitative analysis, SPSS quantitative analysis, research writing, and proofreading articles and reports. I will deliver the highest quality online and offline Excel, R, SPSS, and other spreadsheet solutions within your operational deadlines. I have also compiled many original Excel quantitative and text spreadsheets which solve client’s problems in my research writing career.
I have extensive enterprise resource planning accounting, financial modeling, financial reporting, and company analysis: customer relationship management, enterprise resource planning, financial accounting projects, and corporate finance.
I am articulate in psychology, engineering, nursing, counseling, project management, accounting, finance, quantitative spreadsheet analysis, statistical and economic analysis, among many other industry fields and academic disciplines. I work to solve problems and provide accurate and credible solutions and research reports in all industries in the global economy.
I have taught and conducted masters and Ph.D. thesis research for specialists in Quantitative finance, Financial Accounting, Actuarial science, Macroeconomics, Microeconomics, Risk Management, Managerial Economics, Engineering Economics, Financial economics, Taxation and many other disciplines including water engineering, psychology, e-commerce, mechanical engineering, leadership and many others.
I have developed many courses on online websites like Teachable and Thinkific. I also developed an accounting reporting automation software project for Utafiti sacco located at ILRI Uthiru Kenya when I was working there in year 2001.
I am a mature, self-motivated worker who delivers high-quality, on-time reports which solve client’s problems accurately.
I have written many academic and professional industry research papers and tutored many clients from college to university undergraduate, master's and Ph.D. students, and corporate professionals. I anticipate your hiring me.
I know I will deliver the highest quality work you will find anywhere to award me your project work. Please note that I am looking for a long-term work relationship with you. I look forward to you delivering the best service to you.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
(A) For reaction (20.3), determine (a) The instantaneous rate of reaction at 2400 s and (b) [H 2 O 2 ] at 2450 s. Assume that the instantaneous rate of reaction at 2400 s holds constant for the next...
-
The Tastee Bakery Company supplies a bakery product to many supermarkets in a metropolitan area. The company wishes to study the effect of the height of the shelf display employed by the supermarkets...
-
From the data in Table 14-1 in the text, calculate the overall rate change of first-class postage as measured by the LCI for the following decades: In Table 14.1 (a) The 1970s (1970-1979) (b) The...
-
Roberts Originals Co. (ROC) provides new and unique cases and otherassignments to professors each semester to ensure that students will not be able to find the solutions published online. Due to the...
-
To help you become familiar with the accounting standards, this case is designed to take you to the FASBs Web site and have you access various publications. Access the FASBs Web site at www.fasb.org....
-
A sporting goods manufacturer has decided to expand into a related business. Management estimates that to build and staff a facility of the desired size and to attain capacity operations would cost...
-
Which is not a stage in the perceptual process? (a) attention/selection (b) interpretation (c) follow-through (d) retrieval
-
Ms. Batt owns all the common shares of Batter up Ltd., a Canadian-controlled private corporation, which started operations in 1987. Batter up Ltd. has been quite profitable since the early 1990s. As...
-
Let a and b be real numbers with a
-
The rate of decomposition of gaseous acetaldehyde, CH 3 CHO, to gaseous methane and carbon monoxide is found to increase by a factor of 2.83 when the initial concentration of acetaldehyde is doubled....
-
As shown later in the chapter, for certain reactions the initial and instantaneous rates of reaction are equal throughout the course of the reaction. What must be the shape of the concentrationtime...
-
Calculate the average age of females in the Canadian population in 2014, assuming that the average (midpoint) age of people over 100 is 103. The table below gives the age distribution of the Canadian...
-
08 S On June 30, 2024, Exploration Incorporated signs a lease requiring quarterly payments each year for the next five years. Each of the 20 quarterly payments is $28,969.97, with the first lease...
-
On 6/1/23, Santa wants to review some trading options for his sleds. First, here is some information about his old sled: Purchase Price on 12/31/20: $611,000 Accumulated Depre up to date: $76,000...
-
Good Dog Enterprises sells goods with a warranty. At December 31, 2021 Good Dog has a warranty liability of $2 million and taxable income of $75 million. At December 31, 2020 Good Dog reported a...
-
Suppose that real GDP includes just cars and computers. The United States produces 50 cars and 5 computers, while Japan produces 5 cars and 10 computers, and each country has 10 workers and customers...
-
America's relationship with its mentally ill population continues to suffer due to inadequacies in the country's mental health care system. For the mentally ill in Chicago, the effects of this...
-
Suppose the probability of an IRS audit is 1.7 percent for U.S. taxpayers who le form 1040 and who earned $100,000 or more. (a) What are the odds that such a taxpayer will be audited? (b) What are...
-
Consider the circuit of Fig. 7.97. Find v0 (t) if i(0) = 2 A and v(t) = 0. 1 3 ett)
-
What new rules were enacted under the Sarbanes-Oxley Act to address unethical accounting practices?
-
Stan Kaiser is studying for his next accounting examination. Explain to Stan what he should know about the differences between the income statements for a manufacturing and for a merchandising...
-
Terry Lemay is unclear as to the difference between the balance sheets of a merchandising company and a manufacturing company. Explain the difference to Terry.
-
Here is financial information for Sandhill Inc. December 31, 2022 December 31, 2021 Current assets $124,600 $100,000 Plant assets (net) 394,800 329,000 Current liabilities 120,572 73,000 Long-term...
-
Jones Distributing Company uses the perpetual inventory system and engaged in the following transactions during December of the current year: (Click the icon to view the transactions.) Required 1....
-
5. Given the following cost expenditures (see Table P6.3) for a small bridge job to include direct and indirect charges (but not bank interest): A. Calculate the peak financial requirement. B. Sketch...

Study smarter with the SolutionInn App


