Will the following precipitates form under the given conditions? (a) Pbl(s), from a solution that is 1.05
Question:
Will the following precipitates form under the given conditions?
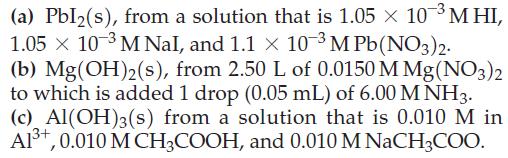
Transcribed Image Text:
(a) Pbl₂(s), from a solution that is 1.05 × 10-³ MHI, 1.05 × 10-3 M Nal, and 1.1 x 10-3 M Pb(NO3)2. (b) Mg(OH)2(s), from 2.50 L of 0.0150 M Mg(NO3)2 to which is added 1 drop (0.05 mL) of 6.00 MNH3. (c) Al(OH)3(s) from a solution that is 0.010 M in Al³+, 0.010 M CH3COOH, and 0.010 M NaCH3COO.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
Precipitate Conditions Will Precipitate PbI2s 1...View the full answer

Answered By

Ali Khawaja
my expertise are as follows: financial accounting : - journal entries - financial statements including balance sheet, profit & loss account, cash flow statement & statement of changes in equity -consolidated statement of financial position. -ratio analysis -depreciation methods -accounting concepts -understanding and application of all international financial reporting standards (ifrs) -international accounting standards (ias) -etc business analysis : -business strategy -strategic choices -business processes -e-business -e-marketing -project management -finance -hrm financial management : -project appraisal -capital budgeting -net present value (npv) -internal rate of return (irr) -net present value(npv) -payback period -strategic position -strategic choices -information technology -project management -finance -human resource management auditing: -internal audit -external audit -substantive procedures -analytic procedures -designing and assessment of internal controls -developing the flow charts & data flow diagrams -audit reports -engagement letter -materiality economics: -micro -macro -game theory -econometric -mathematical application in economics -empirical macroeconomics -international trade -international political economy -monetary theory and policy -public economics ,business law, and all regarding commerce
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
Marx and Engels were two of the most important philosophers in history. Their influence is widely spread around the world to this day. Marx and Engels are deeply critical of the system of life that...
-
Case Study: Quick Fix Dental Practice Technology requirements Application must be built using Visual Studio 2019 or Visual Studio 2017, professional or enterprise. The community edition is not...
-
a) which ionization gives the best sensitivity the analysis of chloramphenicol? b) The compound with highest retention factor? c) What is the resolution between yohimbine and chloramphenicol? d) What...
-
Taxable income and income tax rates for 20092011 for the company have been as follows: Make the journal entry necessary to record any net operating loss (NOL) carryback in2011. Taxable Income Income...
-
The area of a triangle ABC can be calculated by |rAB Ã rAC/2, where rAB and rAC are vectors connecting the vertices A and B and A and C, respectively. Determine the area of the triangle shown...
-
How can team processes be improved?
-
Calculate the moment of inertia of a uniform solid cone about an axis through its center (Fig. 9.40). The cone has mass M and altitude h. The radius of its circular base is R. Axis
-
Sets A, B, and C are subsets of the uni These sets are defined as follows. U={f, g, h, p, q, r, x, y, z A={f, g, q,r} B={g, h, p, r, x} C={q, r,x,y} Find C'U (A'n B). Write your answer in roster form...
-
PbCl 2 (s)is considerably more soluble in HCl(aq) than in pure water, but its solubility in HNO 3 (aq) is not much different from what it is in water. Explain this difference in behavior.
-
To 0.350 L of 0.150 M NH 3 is added 0.150 L of 0.100 M MgCl 2 . How many grams of (NH 4 ) 2 SO 4 should be present to prevent precipitation of Mg(OH) 2 (s)?
-
Dimethylglyoxime [DMG, (CH 3 CNOH) 2 ] is used as a reagent to precipitate nickel ion. Assume that 53.0 g of DMG has been dissolved in 525 g of ethanol (C 2 H 5 OH). (a) What is the mole fraction of...
-
Suppose a triangle has vertices A, B and C and has the following measurements: m/C = 93 AC 39.9 cm a. Start by drawing a diagram of this triangle and labeling the known values. b. What is the length...
-
A space is at a temperature of 75 F (24 C), and the relative humidity is 45 percent. Find (a) the partial pressures of the air and water vapor, (b) the vapor density, and (c) the humidity ratio of...
-
Company A has 1,000 shares of $1 common stock. Owner A owns 60% and Owner B owns 40%. Its Equity Section of its BS looks like this: Common Stock $1,000 Paid in Capital $500 Retained Earnings $500...
-
If total assets are $ 1 2 0 , 0 0 and total liabilities are $ 1 1 2 , 0 0 0 , the amount of stockholder s equity is:
-
The Summit Plummet is an extreme water slideone of the steepest and fastest in the world. Riders drop 36 m from the start until they hit a run-out at the bottom. The slide is steep and slippery, so...
-
Calculate each compound event probability: a. X 10, n = 14, = .95 b. X > 2, n = 5, = .45 c. X 1, n = 10, = .15
-
Express these numbers in standard notation. a. 2.87 10-8 b. 1.78 1011 c. 1.381 10-23
-
Kwun, Inc. purchases 1,000 shares of its own previously issued $5 par common stock for $12,000. Assuming the shares are held in the treasury, what effect does this transaction have on (a) Net income,...
-
The treasury stock purchased in question 14 is resold by Kwun, Inc. for $18,000.What effect does this transaction have on (a) Net income, (b) Total assets, (c) Total paid-in capital, and (d) Total...
-
(a) What are the principal differences between common stock and preferred stock? (b) Preferred stock may be cumulative. Discuss this feature. (c) How are dividends in arrears presented in the...
-
Month Ended December 31, 2016 V flows from operating activities eipts: Collections from customers' yments: For rent For utilities -1400 -80 4800 For salaries -1200 et cash provided (used) by...
-
TB MC Qu. 17-82 (Static) What are the setup costs allocated to... Starbright manufactures child car seats, strollers, and baby swings. Starbright's manufacturing costs are budgeted as follows:...
-
Cash flows from (used for) operating activities-indirect method The income statement disclosed the following items for the year: Depreciation expense Gain on disposal of equipment Net income $38,000...

Study smarter with the SolutionInn App


