Without performing detailed calculations, determine which reaction produces the maximum quantity of O 2 (g) per gram
Question:
Without performing detailed calculations, determine which reaction produces the maximum quantity of O2(g) per gram of reactant.
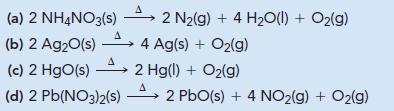
Transcribed Image Text:
2 N₂(g) + 4 H₂O(l) + O₂(g) (a) 2 NH4NO3(s) A (b) 2 Ag₂O(s) → 4 Ag(s) + O₂(g) A (c) 2 HgO(s) 2 Hg(l) + O₂(g) A (d) 2 Pb(NO3)2(s) 2 PbO(s) + 4 NO₂(g) + O₂(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (7 reviews)
The reaction that produces the maximum quantity of O2...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
The main reaction of a charcoal grill is C(s) + O2(g) CO2(g). Which of the statements below are incorrect? Why? a. 1 atom of carbon reacts with 1 molecule of oxygen to produce 1 molecule of CO2. b. 1...
-
Woodalt plc has two automated machine groups X and Y, through which timber is passed in order to produce two models of an item of sports equipment. The models are called ?Traditional? and...
-
16 Cornco produces two products: PS and QT. The sales price for each product and the maximum quantity of each that can be sold during each of the next three months are given in Table 16. Each product...
-
Henries Drapery Service is investigating the purchase of a new machine for cleaning and blocking drapes. The machine would cost $137,320, including freight and installation. Henries estimated the new...
-
Mansfield Company began operations on January 1, 2008, and purchased temporary investments in marketable securities during the year at a cost of $123,000. The end-of-period market value for these...
-
Shanes Shovels produces small, custom earth-moving equipment for landscaping companies. Manufacturing overhead is allocated to work in process using an estimated overhead rate. During April,...
-
Who is protected by the minimum legal capital requirement?
-
The Great Outdoors Clothing Company, a mail-order catalogue operation, contracts with the Federal Parcel Service to deliver all of its orders to customers. As such, Great Outdoors considers Federal...
-
In March, James Electronics had sales of $ 2 , 6 8 0 , 0 0 0 ( 2 6 , 8 0 0 units ) , total variable expenses of $ 1 , 6 0 8 , 0 0 0 , and total fixed expenses of $ 8 9 0 , 0 0 0 . Required: What is...
-
(A) How many grams of magnesium nitride, Mg 3 N 2 , are produced by the reaction of 3.82 g Mg with an excess of N 2 ? (B) How many grams of H 2 are required to produce 1.00 kg methanol, CH 3 OH, by...
-
What mass of CO 2 is formed in the reaction of 4.16 g triethylene glycol, C 6 H 14 O 4 with an excess of O 2 ?
-
An article in the Wall Street Journal noted that many economists believe that GDP data for India are unreliable because most enterprises are tiny and unregistered, and most workers are employed off...
-
I Need Hr project on Employee Engagement What is Employee Engagement and how does it contribute to organizational success? What is the role of HR in improving employee engagement? What are some...
-
In the United States, the Veterans Administration (VA) is tasked with, among other things, providing quality health care for U.S. military veterans. Chronically underfunded, the agency was having...
-
Using the keywords you brainstormed earlier in the module, conduct three separate searches in CQ Researcher - SAGE, Academic Search Ultimate, or another relevant database. When conducting these...
-
TechEx Repair allows local hardware stores to expand their service offerings to their customers by providing an off-site small engine repair service. Customers bring in small engines such as lawn...
-
Purpose: Sometimes we are asked to collaborate with a team of writers. This collaboration can help us to understand how others think differently from us and help us to think more creatively. This...
-
The Reformatsky reaction is an addition reaction in which an organozinc reagent is used instead of a Grignard reagent to attack the carbonyl group of an aldehyde or a ketone. Because the organozinc...
-
The first national bank pays a 4% interest rate compound continuously. The effective annual rate paid by the bank is __________. a. 4.16% b. 4.20% c. 4.08% d. 4.12%
-
You are considering an investment in the common stock of Keller Corp. The stock is expected to pay a dividend of $2 a share at the end of the year (D1 = $2.00). The stock has a beta equal to 0.9. The...
-
What will be the nominal rate of return on a preferred stock with a $100 par value, a stated dividend of 8 percent of par, and a current market price of? (a) $60, (b) $80, (c) $100, and (d) $140?
-
Martell Mining Companys ore reserves are being depleted, so its sales are falling. Also, its pit is getting deeper each year, so its costs are rising. As a result, the companys earnings and dividends...
-
1. Harvey Spectre started a Pearson Hardman on 1 August 2021, a law firm where he will be providing legal services to the companys clients. The following information regarding Pearson Hardman is...
-
On October 20, 2021, a company committed to a plan to sell a division that qualified as a component of the entity according to GAAP regarding discontinued operations and was property classified as...
-
You find that the going rate for a home mortgage with a term of 30 years is 7 % APR. The lending agency says that based on your income, your monthly payment can be at most $ 790 . How much can you...

Study smarter with the SolutionInn App


