Write cell reactions for the electrochemical cells diagrammed here, and use data from Table 19.1 to calculate
Question:
Write cell reactions for the electrochemical cells diagrammed here, and use data from Table 19.1 to calculate E°cell for each reaction.
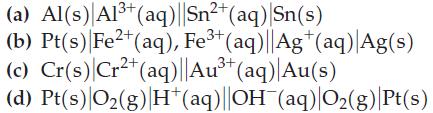
Table 19.1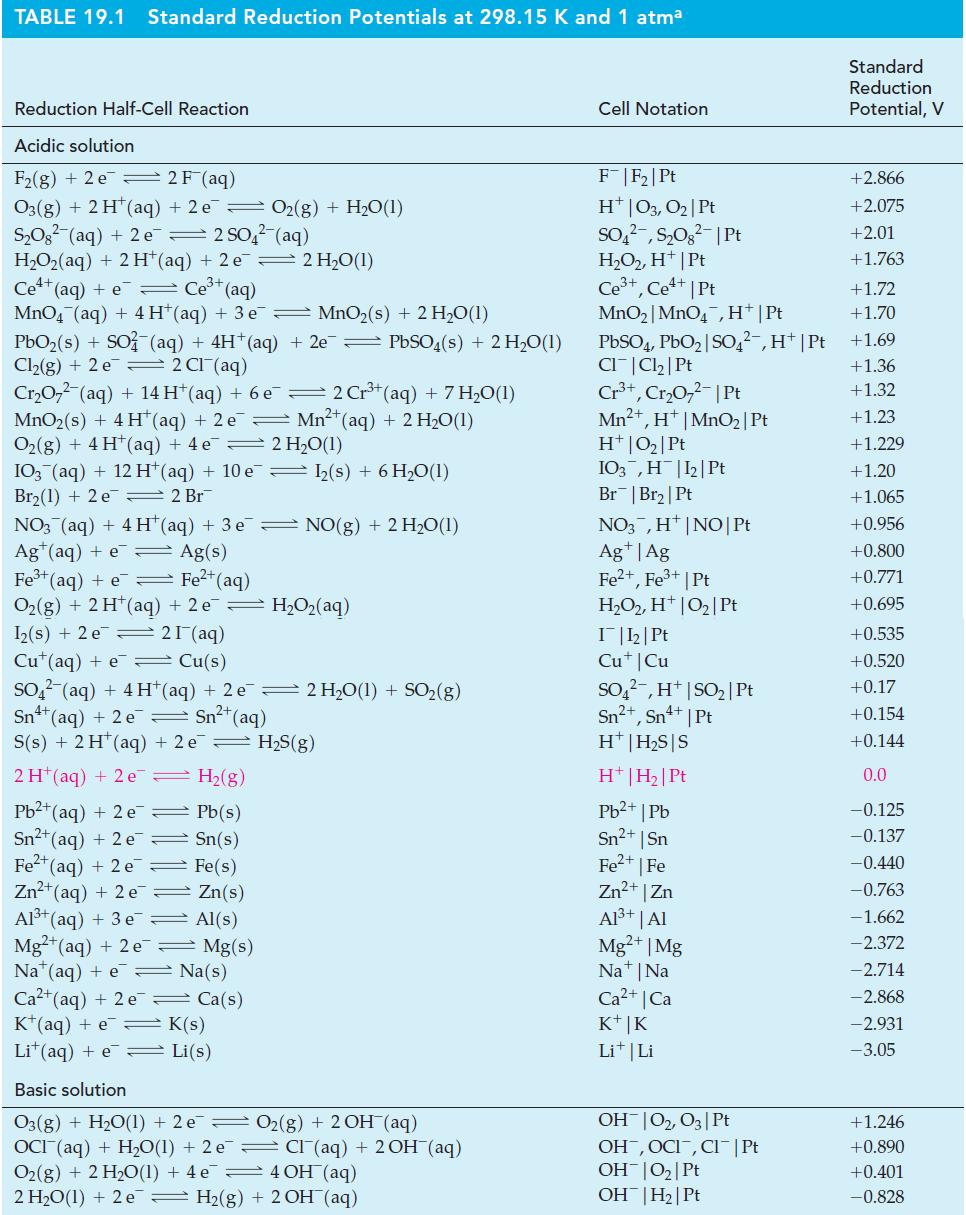
Transcribed Image Text:
2+ (a) Al(s) Al³+ (aq)||Sn²+ (aq)|Sn(s) (b) Pt(s) Fe2+ (aq), Fe³+ (aq)||Ag+ (aq) Ag(s) (c) Cr(s) Cr²+ (aq)||Au³+ (aq)| Au(s) 2+ 3+ (d) Pt(s) O₂(g) H*(aq)||OH¯(aq)|O₂(g) |Pt(s)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)

Answered By

Carly Cimino
As a tutor, my focus is to help communicate and break down difficult concepts in a way that allows students greater accessibility and comprehension to their course material. I love helping others develop a sense of personal confidence and curiosity, and I'm looking forward to the chance to interact and work with you professionally and better your academic grades.
4.30+
12+ Reviews
21+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
Write the half-cell reactions and the balanced chemical equation for the electrochemical cells diagrammed here. Use data from Table 19.1 and Appendix D to calculate E cell for each reaction. Table...
-
(A) Use data from Table 19.1 to predict the probable products when Pt electrodes are used in the electrolysis of KI(aq). Table 19.1 (B) In the electrolysis of AgNO 3 (aq), what are the expected...
-
Use the table below to find: (fog)(-10) = (fof) (11) = X -10 f(x) -1 g(x) 4 -1 -7 -4 -7 11 (gof)(-4)= (gog)(7) = 4 7 15 4 7 -4 11 -4 15 7 -1 15 -10 -7 -10 11
-
93) Clay of the Land is a manufacturer of glazed clay pots. Currently, it produces 300 clay pots per month which it sells through nurseries at a constant price of $5 per pot. Current demand for clay...
-
Discuss the various methods of overcoming blocked channels.
-
Ahi Corporation is one of your clients in Hawaii. The company had a good year last year and owes the IRS $ 100 million, due on March 15. There are no penalties or interest because of the IRS. One of...
-
All sales of Dunns Building Supplies (DBS) are made on credit. Sales ar billed twice monthly, on the tenth of the month for the last half of the prior months sales and on the 20th of the month for...
-
Kobe transfers $500,000 in cash to newly formed Bryant Corporation for 100% of Bryants stock. In the first year of operations, Bryants taxable income before any payments to Kobe is $120,000. What...
-
If the universal set U = {2, 4, 6, 8, 10} and F = {4, 10}, then which set represents F'? {1, 3, 5, 7, 9} {2,6,8} O {4, 6, 8, 10} 5 7 9
-
Use the data in Appendix D to calculate the standard cell potential for each of the following reactions. Which reactions will occur spontaneously? (a) H(g) + F2 (g) (b) Cu(s) + Ba+ (aq) 2 H(aq) + 2 F...
-
Consider the reaction Co(s) + Ni 2+ (aq) Co 2+ (aq) + Ni(s), with E cell = 0.02 V. If Co(s) is added to a solution with [Ni 2+ ] = 1 M, should the reaction go to completion? Explain.
-
In small groups, each member should select an occupation and design a performance management system for the position. Identify what sources would be appropriate for performance feedback and the best...
-
Explain the difference between a voluntary and mandatory change in accounting policy.
-
What does it mean to "recast" the financial statements? Why are financial statements recast?
-
We have had Paige & Gentry as our auditors for many years, havent we, Jane? They have been here since I became president two years ago. Yes, Bob, I have been the CFO for seven years, and they were...
-
A railroad company is trying to decide how to charge its customers for carrying freight. A manager suggests the following would be a good measure upon which to base the price of transporting freight:...
-
If there is doubt about whether a change is a change in policy or a change in estimate, how should it be treated? Why do you think accounting standards have this requirement?
-
The following table gives the seven most common first names among girls born in the United States during 2010 along with their frequencies (in thousands). Name Number of girls (in thousands)...
-
What will be the final value of DI after executing the following piece of code? Execute the instructions dependently one after another. CLD MOU CX,OFOH MOU AX.02874H MOU DI,01000H MOU ES, DI SUB...
-
Columbia Sportswear Company had accounts receivable of $206,024,000 at the beginning of a recent year, and $267,653,000 at year-end. Sales revenues were $1,095,307,000 for the year. What is the...
-
Young Corporation reported income taxes of $340,000,000 on its 2010 income statement and income taxes payable of $277,000,000 at December 31, 2009, and $522,000,000 at December 31, 2010. What amount...
-
Flynn Corporation reports operating expenses of $80,000 excluding depreciation expense of $15,000 for 2010. During the year prepaid expenses decreased $6,600 and accrued expenses payable increased...
-
Using the guidelines covered in Lesson 7, identify which sentence of the three sentences is correct. All new bank customers' qualify for free checking accounts. All new bank customer's qualify for...
-
When it comes to paying state and local taxes in Texas, residents can expect to pay which of the following? Correct Answer(s) income tax Press Space to open property tax Press Space to open sales tax...
-
Which of the following forms or schedules is correct for reporting Oregon itemized deductions? Question 23Select one: a. No additional form needs to be completed, the taxpayer will need to attach...

Study smarter with the SolutionInn App


