Without looking at Table 9.3, arrange the following in order of increasing ionic radius: As 3 ,
Question:
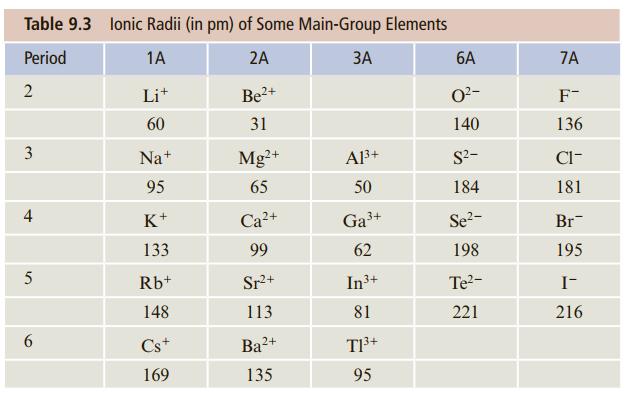
Without looking at Table 9.3, arrange the following in order of increasing ionic radius: As3−, Se2−, Br−. Explain how you arrived at this order.
Transcribed Image Text:
Table 9.3 lonic Radii (in pm) of Some Main-Group Elements Period 1A 2A ЗА 6A 7A 2 Lit 2+ O2- F- 60 31 140 136 3 Na+ Mg?+ A3+ S2- Cl- 95 65 50 184 181 K+ Ca?+ Ga3+ Se?- Br 133 99 62 198 195 5 Rb+ Sr+ In3+ Te?- I- 148 113 81 221 216 Cs+ Ba2+ 169 135 95
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 58% (12 reviews)
Br Se 2 As 3 These ions are members of a...View the full answer

Answered By

Morgan Njeri
Very Versatile especially in expressing Ideas in writings.
Passionate on my technical knowledge delivery.
Able to multitask and able to perform under pressure by handling multiple challenges that require time sensitive solution.
Writting articles and video editing.
Revise written materials to meet personal standards and satisfy clients demand.
Help Online Students with their course work.
4.90+
12+ Reviews
38+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Arrange the following in order of increasing radius and increasing ionization energy. a. N+, N, N- b. Se, Se-, Cl, Cl+ c. Br-, Rb+, Sr2+
-
Without looking at Table 9.3, arrange the following ions in order of increasing ionic radius: Cl, Ca2+, P3. (You may use a periodic table.)
-
Arrange the following in order of increasing ionic radius: Cl, Na+, and S2. Explain this order. (You may use a periodic table.)
-
In Exercises 3336, use possible symmetry to determine whether each graph is the graph of an even function, an odd function, or a function that is neither even nor odd. ------ -2, y CI (0,4) 2 14...
-
On February 4 of a particular year, the spot rate for U.S. dollars ($) expressed in Euros () was $0.7873/. The U.S. interest rate (compounded semiannually) was 5.36 percent, whereas the euro interest...
-
Gouges on a spindle. A tool-and-die machine shop produces extremely high-tolerance spindles. The spindles are 18-inch slender rods used in a variety of military equipment. A piece of equipment used...
-
Why are countries such as Mexico, Brazil and India often considered as ideal manufacturing bases for global corporations? LO.1
-
Consider the following data for the Assembly Division of Fenton Watches Inc. Degree of completion: direct materials, 90%; conversion costs, 40%. Degree of completion: direct materials, 60%;...
-
Miguel and Katrina have 2 children under age 17, have earned income of $75,000. Miguels father lives with them and is considered a dependent. What amount can they claim as a child credit. $______
-
Consider a fully filled tank of semi-circular cross section tank with radius R and width of b into the page, as shown in Fig. P5-11. if the water is pumped out of the tank at flow rate of V = Kh2,...
-
Arrange the following in order of increasing ionic radius: F - , Na + , and N 3- . Explain this order.
-
Arrange the members of each of the following pairs in order of increasing radius and explain the order: a. Ca, Ca 2+ b. P, P 3
-
Find the derivative of the following functions by first expanding the expression. h(x) = (x 2 + 1) 2
-
7. A psychiatrist is testing a new ADHD Medication, which seems to have the potentially harmful side effect of increasing the heart rate. For a sample of 50 clinical study participants whose pulse...
-
Determine the type of engagement that your colleague completed for the client. Justify the selected engagement type for the client. Assess the purpose of each financial statement for the client's...
-
Mills Corporation acquired as a long-term investment $235 million of 8% bonds, dated July 1, on July 1, 2024. Company management has classified the bonds as an available-for-sale investment. The...
-
A force of 28 pounds acts on the pipe wrench shown in the figure below. 18 in. 30 (a) Find the magnitude of the moment about O by evaluating ||OA x F||. (0 0 180) Use a graphing utility to graph the...
-
Module 1 1. There has been a rise in cases of measles in RI. The RI Health Department is wondering if the rate of MMR vaccinations has declined since the start of the COVID-19 pandemic. The...
-
On January 1, 20X1, Mason Manufacturing borrows $500,000 and uses the money to purchase corporate bonds for investment purposes. Interest rates were quite volatile that year and so were the fair...
-
The Ferris wheel in the figure has a radius of 68 feet. The clearance between the wheel and the ground is 14 feet. The rectangular coordinate system shown has its origin on the ground directly below...
-
What are the two factors needed to explain the differences in solubilities of substances?
-
Explain how soap removes oil from a fabric.
-
Consider two hypothetical pure substances, AB(s) and XY(s). When equal molar amounts of these substances are placed in separate 500-mL samples of water, they undergo the following reactions: AB(s) ...
-
Randy (48) takes a $22,000 distribution from his IRA to pay some of his daughter's $28,000 qualified education expenses at an eligible educational institution. His daughter paid $18,000 of her...
-
The takeover specialist chose to use the value derived from dividend discount model, while the directors prefer to use Net Realisable Value approach. Critically discuss the reasoning of each parties...
-
Tony and Suzie graduate from college in May 2021 and begin developing their new business. They begin by offering clinics for basic outdoor activities such as mountain biking or kayaking. Upon...

Study smarter with the SolutionInn App


