(a) By considering Fig. 27.9, suggest a method for the separation of Am from U, Np and...
Question:
(a) By considering Fig. 27.9, suggest a method for the separation of Am from U, Np and Pu.
(b) What would you expect to happen when a solution of NpO2(ClO4)2 in 1Μ HClO4 is shaken with Zn amalgam and the resulting liquid decanted from the amalgam and aerated?
Figure 27.9.
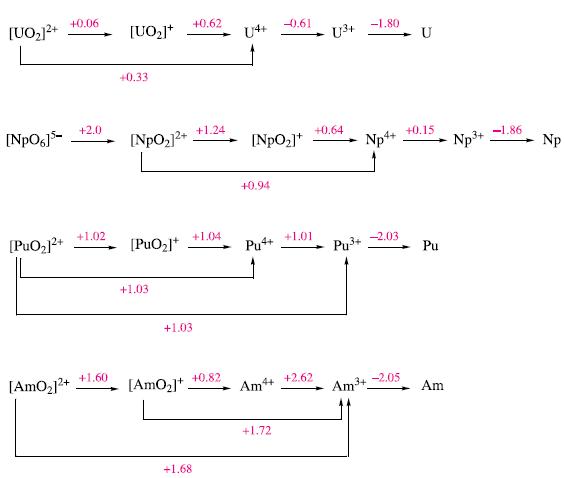
Transcribed Image Text:
[UO₂]²+ [NpO6]5- [PuO₂]²+ [AmO₂]²+ +0.06 +2.0 +1.02 +1.60 [UO₂]+ +0.33 [NpO₂]²+ [PuO₂]* +1.03 +1.03 [AmO₂]+ +0.62 +1.24 +1.04 +1.68 +0.82 [NpO₂]* +0.94 Pu4+ Ĵ Am4+ -0.61 +1.72 +1.01 +2.62 U³+ +0.64 Pu³+ -1.80 Np4+ Am ³+ -2.03 -2.05 U +0.15 Pu Am 3+ Np³+ -1.86 Np
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (5 reviews)
a Method for the separation of Am from U Np and Pu From the given redox potentials standard reduction potentials for various oxidation states of urani...View the full answer

Answered By

User l_1006857
I am a computer science professional with expertise in databases, AI programming, data structures and algorithms, and mathematics. With a strong background in these areas, I possess the knowledge and skills necessary to design and optimize database systems, develop intelligent algorithms and models, and solve complex computational problems. My proficiency in SQL, NoSQL, machine learning techniques, and mathematical concepts equips me to contribute to innovative projects and drive technological advancements.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
What would you expect to happen when chlorine gas, Cl2, at 1 atm pressure is bubbled into a solution containing 1.0 M F and 1.0 M Br at 25oC? Write a balanced equation for the reaction that occurs.
-
In Chapter 7, (Data in Tompkins County Bridges 2014) we found a relationship between the age of a bridge in Tompkins County, New York, and its condition as found by inspection. But we considered only...
-
What would you expect to happen to the risk-free rate and equity returns when a segmented country opens its capital markets to foreign investment?
-
There is a crop that the value p next month is random. The value can be either small p=1 or large P=3 depending on the weather. Half of people are optimists believing the value of the crop will be...
-
Describe how the six elements of a standard unit cost increase cost comparability and understandability.
-
Given the unity feedback system shown in Figure P8.3, where do the following problem parts by first making a second-order approximation. After you are finished with all of the parts, justify your...
-
Assume that on December 31, 20x7, the cost of Rapid Tech Companys portfolio of long-term available-for-sale securities was $1,280,000 and that its market value was $1,200,000. Prepare the entry to...
-
On June 1, 2019, Kris Storey established an interior decorating business, Eco-Centric Designs. During the month, Kris completed the following transactions related to the business: June 1. Kris...
-
\f24 Let f(x) = 2x + Then find the equation of the tangent line to the graph of f(x) at the point (2, 10) in the form y = me + b.If e) = 22:2 51:: + 6, find f'[2). mm = Ii il Use this to find the...
-
Vasovist is the tradename of a Gd(III) complex which was the first intravascular contrast agent (see Box 4.3) approved in the EU for use in magnetic resonance angiography. Interactions between the...
-
(a) If, in an electron-transfer process, there is both electron and ligand transfer between reagents, what can you conclude about the mechanism? (b) Explain why very fast electron transfer between...
-
On January 1, 2008, Phantom Company acquires $200,000 of Spiderman Products, Inc., 9% bonds at a price of $185,589. The interest is payable each December 31, and the bonds mature December 31, 2010....
-
Submit at least 300 word typed self-evaluation in essay form. Please do not submit answers to questions like a survey. Rather, demonstrate critical thinking by providing analysis and evaluation of...
-
Liquid Hair Inc. has two support departments: HR and Admin. The company produces shampoo for women and shampoo for men. HR costs incurred were 35,050$ and Admin costs incurred were 30,100$. HR spent...
-
You plan to deposit $6,100 at the end of each of the next 15 years into an account paying 11.3 percent interest. a. How much will you have in your account if you make deposits for 15 years? (Do not...
-
3.One-quarter of purchases of materials are paid for in the month of purchase.The remainder are paid for two months later.Purchases in the previous two months were:November $108 000; December $106...
-
Read the article You Have an Amazing New Healthcare Innovation, Now What? Legacy DNA. The article advises healthcare innovators to tout the innovative elements of their products or services loudly...
-
Let F 1 and F 2 be d.f.s, and let G be their convolution, G = F 1 * F 2 . Then: (i) If F 1 is absolutely continuous with respect to Lebesgue measure with p.d.f. p 1 , it follows that G is absolutely...
-
Representative data read from a plot that appeared in the paper Effect of Cattle Treading on Erosion from Hill Pasture: Modeling Concepts and Analysis of Rainfall Simulator Data (Australian Journal...
-
Dimethoxyethane (DME) is a polar aprotic solvent often used for S N 2 reactions. Propose a plausible synthesis for DME using acetylene and methyl iodide as your only sources of carbon atoms....
-
Using compounds that possess no more than two carbon atoms, propose a plausible synthesis for the following compound.
-
Assign an IUPAC name for each of the following compounds. a. b. c. d. e. f. g. SH
-
Analyze the standard costing system employed by Tesla, Inc. For a specific product, the standard cost per unit is $50, comprising $30 for direct materials and $20 for direct labor. During the last...
-
Transfer Pricing at Microsoft Microsoft is evaluating its transfer pricing strategy. Values: Item Amount Production Cost $80 per unit Profit Margin 30% Requirements: Determine the minimum transfer...
-
Luna was recruited to serve as the Chief Financial Officer (CFO) of Alpha Corporation, a publicly-traded company in the healthcare industry, effective March 1, 2022. For the fiscal year 2023, Alpha...

Study smarter with the SolutionInn App


