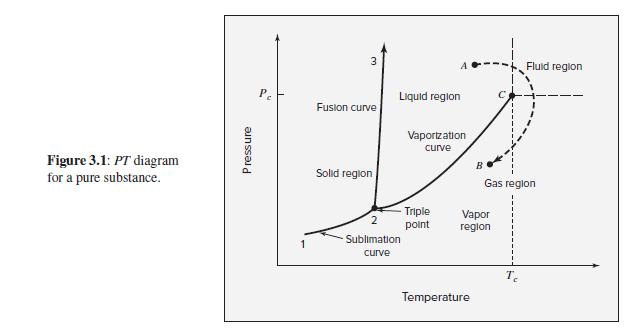
As suggested by Fig. 3.1, the slope of the sublimation curve at the triple point is generally
Question:
As suggested by Fig. 3.1, the slope of the sublimation curve at the triple point is generally greater than that of the vaporization curve at the same state. Rationalize this observation. Note that triple-point pressures are usually low; hence assume for this exercise that Δ Zsv ≈ Δ Zlv ≈ 1.
Fig. 3.1
Transcribed Image Text:
Fluid reglon Liquid region Fusion curve Vaportzation curve Figure 3.1: PT diagram for a pure substance. Solid region Gas region Triple Vapor region point Sublimation curve Temperature Pressure
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (8 reviews)
The slope of the sublimation curve at the triple point is generally greater than that of the vaporiz...View the full answer

Answered By

BillClinton Muguai
I have been a tutor for the past 5 years. I have experience working with students in a variety of subject areas, including computer science, math, science, English, and history. I have also worked with students of all ages, from elementary school to college. In addition to my tutoring experience, I have a degree in education from a top university. This has given me a strong foundation in child development and learning theories, which I use to inform my tutoring practices.
I am patient and adaptable, and I work to create a positive and supportive learning environment for my students. I believe that all students have the ability to succeed, and it is my job to help them find and develop their strengths. I am confident in my ability to tutor students and help them achieve their academic goals.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Introduction To Chemical Engineering Thermodynamics
ISBN: 9781259696527
8th Edition
Authors: J.M. Smith, Hendrick Van Ness, Michael Abbott, Mark Swihart
Question Posted:
Students also viewed these Engineering questions
-
Point a in Fig is maintained at a constant potential of 400 V. above ground. (See Problem) (a) What is the reading of a voltmeter with the proper range and with resistance 5.00 X 104 Ω when...
-
Assume the same data as in Appendix 1 Exercise 12-19, Except that the current interest rate is 12%. In Appendix 1 Exercise 12-19, On January 1, 2014, you win $75,000,000 in the state lottery. The...
-
At what point on the curve y = 1 + 2ex 3x is the tangent line parallel to the line 3x y = 5? Illustrate by graphing the curve and both lines.
-
The objective of this problem is to design and develop a program for Huffman coding algorithm. The discrete source has an alphabet X = {x1, x2, x3, x4, x5, x6, x7, x8, x9} with corresponding...
-
List the information required for accurate meeting minutes.
-
In your opinion, did the apparent mistakes made by the PwC auditors in auditing Take- Twos receivables and reserve for sales returns involve negligence on their part? Would you characterize the...
-
A colleague has collected data from a sample of 80 students. He presents you with the following output from the statistical analysis software: LO9 Explain what this tells you about undergraduate and...
-
If Webster had made the chowder herself from a recipe that she had found on the Internet, could she have successfully brought an action against its author for a breach of the implied warranty of...
-
10. 11. 10 On January 1, Year 1, Bell Corp. issued $180,000 of 10-year, 6 percent bonds at their face amount, Interest is payable on December 31 of each year with the first payment due December 31,...
-
1. What types of conflict is the team experiencing in the challenge video? 2. What aspects of the negotiation process would best resolve the conflict and why would this work? 3. What conflict...
-
An ideal gas with constant heat capacities enters a converging/diverging nozzle with negligible velocity. If it expands isentropically within the nozzle, show that the throat velocity is given by:...
-
Real-gas behavior for turbomachinery is sometimes empirically accommodated through the expression W = Z W ig , where W ig is the ideal-gas mechanical power and Z is some suitably defined average...
-
How many days are in the billing cycle? ACCOUNT INFORMATION Account Number TRANSACTIONS 9 MAY 12 MAY 15 MAY 18 MAY 21 MAY SUMMARY 3291684271 Fanelli Furs 594683219 Brooklyn Pets 7677095385 Maple...
-
You have been employed as a systems analyst in the information systems organization of a medium-sized consumer goods manufacturer for three years. You are quite surprised when your manager offers you...
-
For your initial post, address the following: First, introduce yourself to the class by sharing a bit about yourself, such as your preferred name or pronouns, where you are from, what your major is,...
-
Question 8 : Consider the technology of Solar Panels. Which stage of the technology life cycle S curve is this technology in. Justify why ? Question 9 : The standard Product Life Cycle has 5 stages...
-
At Benihana restaurant a man wrenched his neck while ducking a piece of flying shrimp, requiring treatment by several doctors. By that summer, doctors determined surgery was necessary to treat...
-
You have just come into an inheritance of $25,000 from a distant relative, and you want to invest it for the long term. Provide an investment portfolio that includes five different stocks. Report the...
-
At the beginning of 2019, Brent Company amended its defined benefit pension plan. The amendment entitled five active participating employees to receive increased future benefits based on their prior...
-
At Glass Company, materials are added at the beginning of the process and conversion costs are added uniformly. Work in process, beginning: Number of units Transferred - in costs Direct materials...
-
The Reynolds number Re is a dimensionless group that characterizes the intensity of a flow. For large Re, a flow is turbulent; for small Re, it is laminar. For pipe flow, Re uD/, where D is pipe...
-
A list of common unit operations follows: (a) Single-pipe heat exchanger (b) Double-pipe heat exchanger (c) Pump (d) Gas compressor (e) Gas turbine (f) Throttle valve (g) Nozzle Develop a simplified...
-
A solid body at initial temperature T 0 is immersed in a bath of water at initial temperature T w0 . Heat is transferred from the solid to the water at a rate Q = K (T w T) , where K is a constant...
-
To fund your dream around-the-world vacation, you plan to save $1,300 per year for the next 14 years starting one year from now. If you can earn an interest rate of 5.83 percent, how much will you...
-
On NSE (Indian stock exchange), shares of ICICI Bank trade for 935 rupees. If the spot exchange rate is USD 0.012, what is the no-arbitrage USD price of ICICI Bank ADR? Assume that transactions costs...
-
Income Statement Balance Sheet Balance Sheet Additional Financial Information 1. Market price of Ranfield's common stock: $90.44 at December 31, 2024, and $58.35 at December 31, 2023. 2. Common...

Study smarter with the SolutionInn App


