(a) Derive Equation 4.131 from Equation 4.130. Use a test function; otherwise youre likely to drop some...
Question:
(a) Derive Equation 4.131 from Equation 4.130. Use a test function; otherwise you’re likely to drop some terms.
(b) Derive Equation 4.132 from Equations 4.129 and 4.131. Use Equation 4.112.
Equation 4.112

Equations 4.129

Equations 4.130

Equations 4.131
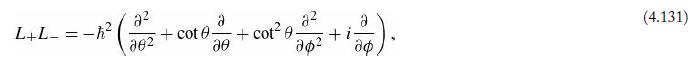
Equations 4.132
Transcribed Image Text:
(4.112) • ²74 ± ²7+7+7=z1
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 42% (7 reviews)
a b Equation 4129 LLf held LL leofe ofic10 icot 0ie 02 f 802 2 20 2 ...View the full answer

Answered By

Ann Wangechi
hey, there, paying attention to detail is one of my strong points, i do my very best combined with passion. i enjoy researching since the net is one of my favorite places to be and to learn. i am a proficient and versatile blog, article academic and research writing i possess excellent English writing skills, great proof-reading. i am a good communicator and always provide feedback in real time. i'm experienced in the writing field, competent in computing, essays, accounting and research work and also as a Database and Systems Administrator
4.90+
151+ Reviews
291+ Question Solved
Related Book For 

Introduction To Quantum Mechanics
ISBN: 9781107189638
3rd Edition
Authors: David J. Griffiths, Darrell F. Schroeter
Question Posted:
Students also viewed these Engineering questions
-
Use Equation (7.35) (page 541) to derive the formula Equation (7.35) 1 (x) i=1
-
Use Equation (7.35) (page 545) to derive the formula Equation (7.35) o? =o? Po .2 E (x; X)? in n i31 +
-
Use Equation (7.34) (page 545) to derive the formula Equation (7.34) .2 E (x; T)? B1 i=1 ||
-
Villaverde Company insures the life of its president for P8,000,000, the corporation being the beneficiary of an ordinary life policy. The premium is P200,000. The policy is dated January 1, 2010....
-
Vaneeta Construction Ltd. began operations in 2014. Construction activity for the first year follows. All contracts are with different customers, and any work remaining at December 31, 2014, is...
-
Describe the flow of costs through the inventory accounts of a manufacturing firm.
-
Use Laplace transforms to solve the following initial value problems. Where possible, describe the solution behavior in terms of oscillation and decay. a. \(y^{\prime \prime}-5 y^{\prime}+6 y=0,...
-
On October 1, White Way Stores Inc. is considering leasing a building and purchasing the necessary equipment to operate a retail store. Alternatively, the company could use the funds to invest in $...
-
Suppose that FDU Financial Inc. issued a bond with 10 years until maturity, a face value of $1000, and a coupon rate of 11% (annual payments). The yield to maturity on this bond when it was issued...
-
Determine VD for the fixed-bias configuration of Fig. 7.83. 20 V 2.2 k DSS 1
-
(a) Prove that for a particle in a potential V(r) the rate of change of the expectation value of the orbital angular momentum L is equal to the expectation value of the torque: where (This is the...
-
Consider the earthsun system as a gravitational analog to the hydrogen atom. (a) What is the potential energy function (replacing Equation 4.52)? (Let m E be the mass of the earth, and M the mass of...
-
Two connected wires with linear mass densities that are related by 1 = 32 are under the same tension. When the wires oscillate at a frequency of 120 Hz waves of wavelength 10 cm travel down the first...
-
Police Officer Jones, in full uniform, was walking the midnight shift in a high-crime area at approximately 2:00 AM and is approached by a civilian who told Officer Jones she had been robbed and...
-
You are writing a legal memorandum and want to include the following list. Which numbering system would be your best option? The three requirements of standing are 1) injury in fact, 2) causation,...
-
We've been reviewing the duty to treat and the duty of care. I'd like to take a couple of minutes to review that section so that hopefully you have it all down. And then we'll move on to the issue of...
-
In the below circuit there is a 4.88 volts battery and three resistors with the following values: R 1 = 6.42 ohms, R 2 = 5.85 ohms, and R 3 = 4.81 ohms. Find the equivalent resistance (in ohms) for...
-
Leo and Elizabeth were getting married and planned a wedding reception for 150 people. They entered a contract with Snuffy's, a banquet hall, to hold the reception between 6:00 p.m. and 11:00 p.m. on...
-
Determine the last digit in 355.
-
On January 2, 20X3, Sheldon Bass, a professional engineer, moved from Calgary to Edmonton to commence employment with Acco Ltd., a large public corporation. Because of his new employment contract,...
-
(a) What value of absorbance corresponds to 45.0% T? (b) If a 0.010 0 M solution exhibits 45.0% T at some wavelength, what will be the percent transmittance for a 0.020 0 M solution of the same...
-
(a) A 3.96 10 4 M solution of compound A exhibited an absorbance of 0.624 at 238 nm in a 1.000-cm cuvet; a blank solution containing only solvent had an absorbance of 0.029 at the same wavelength....
-
Ammonia can be determined spectrophotometrically by reaction with phenol in the presence of hypochlorite (OCl - ): 4.37-mg sample of protein was chemically digested to convert its nitrogen into...
-
For the reaction Fe (s) + NiCl2 (aq) ? 2 Ni (s) + FeCl2 (aq), identify: 1) the oxidation number of Ni (s) 2) the oxidation number of Fe in the FeCl2 (aq) 3) the oxidation number of Cl in the FeCl2...
-
All of these choices contribute to improving your processes. But which is the most important factor? Select an answer:- a.) Adjust operating practices and procedures. b.) Have total involvement in...
-
Information regarding Barrymore Furniture Company's March Quarter is given Direct labor $450.000 Beginning work in process inventory $320,000 Ending work in process inventory $310,000 Manufacturing...

Study smarter with the SolutionInn App


