Ammonia can be determined spectrophotometrically by reaction with phenol in the presence of hypochlorite (OCl - ):
Question:
Ammonia can be determined spectrophotometrically by reaction with phenol in the presence of hypochlorite (OCl-):
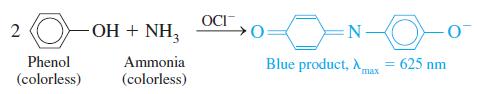
4.37-mg sample of protein was chemically digested to convert its nitrogen into ammonia and then diluted to 100.0 mL. Then 10.0 mL of the solution were placed in a 50-mL volumetric flask and treated with 5 mL of phenol solution plus 2 mL of sodium hypochlorite solution. The sample was diluted to 50.0 mL, and the absorbance at 625 nm was measured in a 1.00-cm cuvet after 30 min. For reference, a standard solution was prepared from 0.010 0 g of NH4Cl (FM 53.49) dissolved in 1.00 L of water. Then 10.0 mL of this standard were placed in a 50-mL volumetric flask and analyzed in the same manner as the unknown. A reagent blank was prepared by using distilled water in place of unknown.
Absorbance | |
Sample | at 625 nm |
Blank | 0.140 |
Reference | 0.308 |
Unknown | 0.592 |
(a) Calculate the molar absorptivity of the blue product.
(b) Calculate the weight percent of nitrogen in the protein.
Step by Step Answer:






