If we continuously compress a real gas in a cylinder with a movable piston while keeping the
Question:
If we continuously compress a real gas in a cylinder with a movable piston while keeping the container at a constant temperature in an ice bath:
(a) What will eventually happen to the gas and why?
(b) How well will the ideal gas law apply to the real gas shown when it is close to doing what you said it will do in part (a)?
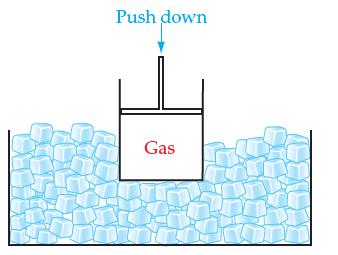
Transcribed Image Text:
Push down Gas
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (6 reviews)
a If we continuously compress a real gas in a cylinder with a movable piston while keeping the container at a constant temperature in an ice bath the ...View the full answer

Answered By

Aysha Ali
my name is ayesha ali. i have done my matriculation in science topics with a+ . then i got admission in the field of computer science and technology in punjab college, lahore. i have passed my final examination of college with a+ also. after that, i got admission in the biggest university of pakistan which is university of the punjab. i am studying business and information technology in my university. i always stand first in my class. i am very brilliant client. my experts always appreciate my work. my projects are very popular in my university because i always complete my work with extreme devotion. i have a great knowledge about all major science topics. science topics always remain my favorite topics. i am also a home expert. i teach many clients at my home ranging from pre-school level to university level. my clients always show excellent result. i am expert in writing essays, reports, speeches, researches and all type of projects. i also have a vast knowledge about business, marketing, cost accounting and finance. i am also expert in making presentations on powerpoint and microsoft word. if you need any sort of help in any topic, please dont hesitate to consult with me. i will provide you the best work at a very reasonable price. i am quality oriented and i have 5 year experience in the following field.
matriculation in science topics; inter in computer science; bachelors in business and information technology
_embed src=http://www.clocklink.com/clocks/0018-orange.swf?timezone=usa_albany& width=200 height=200 wmode=transparent type=application/x-shockwave-flash_
4.40+
11+ Reviews
14+ Question Solved
Related Book For 

Introductory Chemistry Atoms First
ISBN: 9780321927118
5th Edition
Authors: Steve Russo And Michael Silver
Question Posted:
Students also viewed these Sciences questions
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
The Crazy Eddie fraud may appear smaller and gentler than the massive billion-dollar frauds exposed in recent times, such as Bernie Madoffs Ponzi scheme, frauds in the subprime mortgage market, the...
-
A(n) ________ is a short-lived fashion. Relatively few people adopt this product, but it can spread quickly. Question content area bottom Part 1 A. fad B. creolization C. aspirational reference D....
-
Ivy Enterprises Inc. manufactures bathroom fixtures. The stockholders equity accounts of Ivy Enterprises Inc., with balances on January 1, 2010, are as follows: The following selected transactions...
-
What is your best estimate of the percent change in urinary protein based on the data in Table 8.16? Renal Disease Ten patients with advanced diabetic nephropathy (kidney complications of diabetes)...
-
C14.3. What is thedifference between anSF2andan SF3 forecast?
-
Z-Bar Pastures is a 160-hectare farm on the outskirts of Swift Current, Saskatchewan, specializing in the boarding of brood mares and their foals. A recent economic downturn in the thoroughbred...
-
A payday loan company charges 9 percent interest for a two - week period. What would be the annual interest rate from that company?
-
You are trapped on a desert island without any reference books, and you need the value of the ideal gas constant R. Luckily, you stumble upon the following tank of helium (see below). Use what you...
-
Consider two balloons, both in the same room and both exactly the same volume. Consider also the balanced reaction between these gases. If we allow the gases from these balloons to mix and react,...
-
In 2014, Firm Z elected to expense the $8,000 cost of a machine, which it reported as the only item of equipment placed in service that year. This year, the IRS audited Firm Zs 2014 return and...
-
Refer to the information presented in M7-9. Suppose that Juanita has developed a rectangular, medium-size ceramic pot. It requires 3 hours of kiln time; however, two medium-size pots can fit m the...
-
Eclipse Company manufactures a variety of sunglasses. Production information for its most popular line, the Total Eclipse (TE), follows: Suppose that Eclipse has been approached about making a...
-
Sunblocker Corp. makes several varieties of beach umbrellas and accessories. It has been approached about producing a special order for custom umbrellas. The special-order umbrellas with the Randolph...
-
Sunblocker Corp. is considering eliminating a product from its Happy Sand line of beach umbrellas. This collection is aimed at people who spend time on the beach or have an outdoor patio near the...
-
Suppose that annual demand for a certain item has decreased dramatically this year, although the store that stocks this item has not updated its inventory policy, so the store is still using the same...
-
Let Un-1(x) be defined as in Exercise 6 for n > 1 and define U-1(x) = 0. Show that (a) Tn(x) = Un(x) - xUn-1(x), for n > 0 (b) Un{x) = 2xUn-1(x) - Un-2(x), for n > 1
-
Tanaka Company's cost and production data for two recent months included the following: March April Production (units).........300................600 Rent.....................$1,800............$1,800...
-
The circuit in Fig. 2.134 is to control the speed of a motor such that the motor draws currents 5 A, 3 A, and 1 A when the switch is at high, medium, and low positions, respectively. The motor can be...
-
Find R ab in the four-way power divider circuit in Fig. 2.135 . Assume each R = 4 . ww- ww- ER ER R. :R
-
Repeat Prob. 2.75 for the eight-way divider shown in Fig. 2.136 . Prob 2.75 Find R ab in the four-way power divider circuit in Fig. 2.135. Assume each R = 4 Ω. inIim LinLw Lui bo ww- ww-...
-
Docs Auto Body has budgeted the costs of the following repair time and parts activities for 2009: Doc's budgets 6,000 hours of repair time in 2009. A profit margin of $7 per labour hour will be added...
-
QUESTION 28 In a perpetual inventory system, the cost of inventory sold is: Debited to accounts receivable. Debited to cost of goods sold. O Not recorded at the time goods are sold. O Credited to...
-
The following financial statements and additional information are reported. IKIBAN INC. Comparative Balance Sheets June 30, 2019 and 2018 2019 2018 $105,709 69,500 66,800 4,700 246,700 127,eee...

Study smarter with the SolutionInn App


