Sulfuric acid is the single most important industrial chemical. About 40 million tons are manufactured each year.
Question:
Sulfuric acid is the single most important industrial chemical. About 40 million tons are manufactured each year. Elemental sulfur is converted to sulfuric acid by the Contact Process as follows:
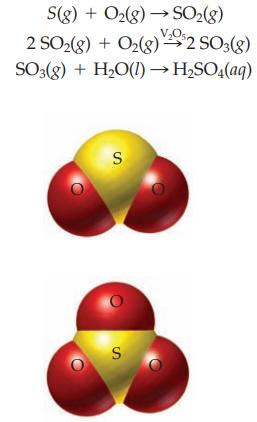
Starting with 1.00 kg of sulfur and excess oxygen gas, calculate:
(a) The mass of sulfur trioxide produced
(b) The STP volume of sulfur trioxide produced
(c) The mass of sulfuric acid produced assuming a 55.0% process yield.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

Introductory Chemistry Concepts And Critical Thinking
ISBN: 9780321804907
7th Edition
Authors: Charles Corwin
Question Posted:





