Figure 4-26 shows an energy loss spectrum for He measured in an apparatus such as that shown
Question:
Figure 4-26 shows an energy loss spectrum for He measured in an apparatus such as that shown in Figure 4-24a. Use the spectrum to construct and draw carefully to scale an energy-level diagram for He.
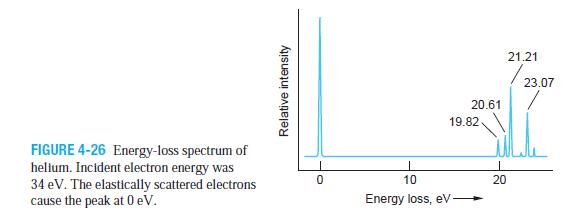
Figure 4-24(a)
Transcribed Image Text:
FIGURE 4-26 Energy-loss spectrum of helium. Incident electron energy was 34 eV. The elastically scattered electrons cause the peak at 0 eV. Relative intensity 0 20.61 19.82 10 Energy loss, eV 20 21.21 23.07
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 85% (7 reviews)
23 22 21 20 ...View the full answer

Answered By

YOGENDRA NAILWAL
As I'm a Ph.D. student, so I'm more focussed on my chemistry laboratory. I have qualified two national level exams viz, GATE, and NET JRF (Rank 68). So I'm highly qualified in chemistry subject. Also, I have two years of teaching experience in this subject, which includes college teacher as well as a personal tutor. I can assure you if you hire me on this particular subject, you are never going to regret it.
Best Regards.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Physics questions
-
An air wedge such as that shown in Fig. 24.30 can be used to measure small dimensions, such as the diameter of a thin wire. (a) If the top glass plate is illuminated with monochromatic light, the...
-
Use the data to construct a 95% confidence interval estimate of the number of credit hours taken by the population of full-time students. Write a brief statement that interprets the result. The...
-
For a finite slope such as that shown in Figure 16.10, assume that the slope failure would occur along a plane (Culmanns assumption). Find the height of the slope for critical equilibrium. Given: ' =...
-
Find the derivative of the function. y = e x-4
-
A study by researchers at the University of Maryland addressed the question of whether the mean body temperature of humans is 98.6F. The results of the study by P. Mackowiak et al. appeared in the...
-
The basic principles of learning that affect training success, such as meaningfulness, practice, overlearning, and feedback
-
When costing profit is Rs 13,500 and a charge in lieu of rent is Rs 2,000, then the financial profit should be (a) Rs 13,500 (b) Rs 15,500 (c) Rs 11,500 (d) None of these
-
The following defined pension data of Doreen Corp. apply to the year 2014. Defined benefit obligation, 1/1/14 (before amendment) .... $560,000 Plan assets, 1/1/14 ................... 546,200 Pension...
-
ant 116 Second Evaluation Chapter 12 am Valuation Chapter 12 and 13(First Part Take Hom 13. Barton and Fallows form a part accounts receivable with a face amount depreciation of $100,000. The...
-
Curve Fitting with Constraints. A banks economist has been interested in developing a production function for the bank. The model recognizes three explanatory variables, which are measures of...
-
A sample of hydrogen atoms are all in the n = 5 state. If all the atoms return to the ground state, how many different photon energies will be emitted, assuming all possible transitions occur? If...
-
In a sample that contains hydrogen, among other things, four spectral lines are found in the infrared with wavelengths 7460 nm, 4654 nm, 4103 nm, and 3741 nm. Which one does not belong to a hydrogen...
-
If U.S. residents take funds out of their checking accounts and put them into time deposits, explain what happens, if anything, to the size of M1, M2, and the monetary base?
-
The adjusted trial balance columns of a worksheet for Levitt Corporation are shown below. The worksheet is prepared for the year ended December 31, Complete the worksheet by (a) entering the adjusted...
-
Derive the commutator $\left[Q_{i}, Q_{j} ight]=i \epsilon_{i j k} Q_{k}$ for the charge defined in Eq. (33.4). Use the charge (33.4) to write the commutator, displaying explicit matrix indices...
-
Verify that the potential $V(\pi, \sigma)$ can be written as Eq. (33.11), and that if $\epsilon=0$ and the symmetry is implemented in the Wigner mode the masses for the $\pi$ and $\sigma$ fields are...
-
Figure 5.7 shows a number of yield curves at various points in time. Go to www.treasury.gov, and in the Resource Center at the top of the page click on Data and Charts Center. Find the Treasury yield...
-
The number of vacation days used by a sample of 20 employees in a recent year In Exercises 2326, use technology to draw a box-and-whisker plot that represents the data set. 3 9 2 17 5 3 2 2 6 4 0 10...
-
Verify that integrating the complex function of Example A.5 along a simple closed curve C 3 , enclosing all three singularities, yields Az)dz= 2ni(1 sin 1)
-
Maria Castigliani is head of the purchasing department of Ambrosiana Merceti, a medium-sized construction company. One morning she walked into the office and said, The main problem in this office is...
-
Two wires are tied to the 300 g sphere shown in FIGURE P8.46. The sphere revolves in a horizontal circle at a constant speed of 7.5 m/s. What is the tension in each of the wires? FIGURE P8.46 1.0 m...
-
Two wires are tied to the 300 g sphere shown in FIGURE P8.46. The sphere revolves in a horizontal circle at a constant speed of 7.5 m/s. What is the tension in each of the wires? FIGURE P8.46 1.0 m...
-
Two wires are tied to the 2.0 kg sphere shown in FIGURE P8.45. The sphere revolves in a horizontal circle at constant speed. a. For what speed is the tension the same in both wires?b. What is the...
-
help me A 35% discount on 3 smart phone amounts to $385. What is the phone's list price? Answer =$ (rounded to the nearest cent)
-
What effect is there on the income statement and balance sheet when an expense is left too long as a liability
-
Which of the following programs covers custodial care? A HMOs B Medicare Part B C PPOs D Medicare Part A E Medicaid

Study smarter with the SolutionInn App


