Using the data for ionic and metallic crystals from Table 10-1, (a) Graph cohesive energy versus melting
Question:
Using the data for ionic and metallic crystals from Table 10-1,
(a) Graph cohesive energy versus melting point and put the best straight line through the points.
(b) Determine the cohesive energies of cobalt, silver, and sodium, whose melting temperatures are 1495°C, 962°C, and 98°C, respectively. (The measured values are cobalt 4.43 eV, silver 2.97 eV, and sodium 1.13 eV.)
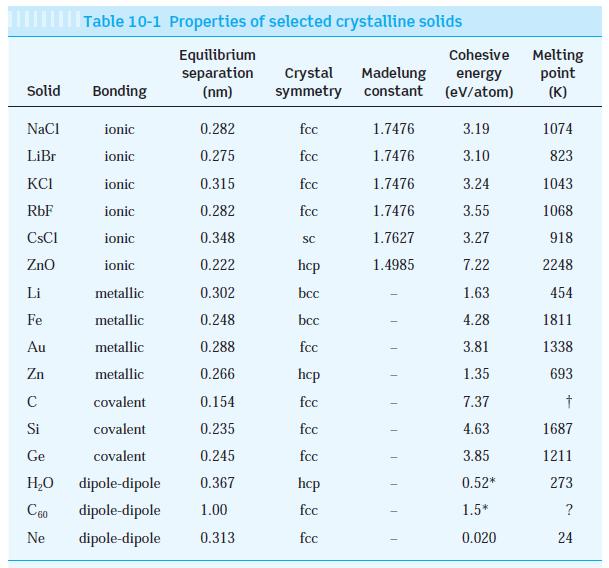
Transcribed Image Text:
Solid NaCl LiBr KCI RbF ionic 0.282 ionic 0.275 ionic 0.315 ionic 0.282 ionic 0.348 ionic 0.222 metallic 0.302 metallic 0.248 metallic 0.288 metallic 0.266 covalent 0.154 covalent 0.235 Ge covalent 0.245 H₂O dipole-dipole 0.367 C60 dipole-dipole 1.00 Ne dipole-dipole 0.313 CsCl ZnO Table 10-1 Properties of selected crystalline solids Equilibrium separation (nm) Li Fe Au Zn C Si Bonding Crystal Madelung symmetry constant fcc fcc fcc fcc SC hcp bcc bcc fcc hcp fcc fcc fcc hcp fcc fcc 1.7476 1.7476 1.7476 1.7476 1.7627 1.4985 T 1 T Cohesive Melting energy (eV/atom) point (K) 3.19 3.10 3.24 3.55 3.27 7.22 1.63 4.28 3.81 1.35 7.37 4.63 3.85 0.52* 1.5* 0.020 1074 823 1043 1068 918 2248 454 1811 1338 693 † 1687 1211 273 ? 24
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (9 reviews)
a b Noting that the melting points are in kelvins on the gr...View the full answer

Answered By

Sumit kumar
I am an experienced online essay writer with a thorough understanding of any curriculum.and subject expert at Chegg for mathematics, CS subjects..
4.90+
5+ Reviews
13+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Physics questions
-
Using the data for Impeccable Travel Service shown in Practice Exercise 1-4A, prepare a statement of owners equity for the current year ended November 30, 2010. Charly Maves invested an additional...
-
Using the data for Express Travel Service shown in Practice Exercise 1-4B, prepare a statement of owners equity for the current year ended June 30, 2010. Janis Paisley invested an additional $30,000...
-
Using the data for Herat Travel Service shown in Practice Exercise 1-4A, prepare a statement of owners equity for the current year ended June 30, 2008. Lola Stahn invested an additional $20,000 in...
-
In an attempt to prepare propylbenzene, a chemist alkylated benzene with 1-chloropropane and aluminum chloride. However, two isomeric hydrocarbons were obtained in a ratio of 2:1, the desired...
-
For Exercises, write the distribution for the formula and determine whether it is a probability distribution. P(X) = X / 6 for X = 3, 4, 7
-
You are caught in the middle of a dilemma. You have been subpoenaed to be a witness in a work-related sexual harassment case involving your boss and a coworker. On many occasions, you heard your boss...
-
If you heard from an employee of a local bank that its a tradition here for us to stand up and defend the bank when someone criticizes it, you could assume that the bank employees had strong...
-
Rhodes parked his car in the self-service park-and-lock lot of Pioneer Parking Lot, Inc. The ticket that he received fromthe ticketmeter stated the following: NOTICE. THIS CONTRACT LIMITS OUR...
-
An organization has members who possess IQs in the top 4% of the population. If IQs are normally distributed, with a mean of 105 and a standard deviation of 15, what is the minimum IQ required for...
-
The unadjusted trial balance of PS Music as of July 31, 20Y9, along with the adjustment data for the two months ended July 31, 20Y9, are shown in Chapter 3. Based upon the adjustment data, the...
-
The crystal structure of KCl is the same as that of NaCl. (a) Calculate the electrostatic potential energy of attraction of KCl, assuming that r 0 is 0.314 nm. (b) Assuming that n = 9 in Equation...
-
The microwave spectrum of CO has lines at 0.86 mm, 1.29 mm, and 2.59 mm. (a) Compute the photon energies and carefully sketch the energy-level diagram that corresponds. What molecular motion produces...
-
Let f, g be real functions j and for each x Dom (f) Dom (g) define (f g)(x) :=max{f(x),g(x)} and (f g)(x) := mm{f (x), g(x)}. a) Prove that and for all x Dom (f) Dom (g). b) Prove that if...
-
The total electric flux from a cubical box of side. 13.0 cm is 1.85x10 Nm/C. What charge is enclosed by the box?
-
Kaylyn Company purchased an intangible asset for $ 4 5 0 , 0 0 0 on January 1 of Year 1 . On January 1 of Year 2 , the asset was evaluated to determine whether it was impaired. As of January 1 of...
-
A neutron in a reactor makes an elastic head-on collision with the nucleus of an atom initially at rest. Assume: The mass of the atomic nucleus is about 14.9 the mass of the neutron. What fraction of...
-
johnnys Lunches is considering purchasing a new, energy-efficient grill. The grill will cost $43,000 and will be depreciated straight-line over 3 years. It will be sold for scrap metal after 5 years...
-
You are in the board of management for an organization that is going through challenges due to harsh economic conditions. A board meeting has been set up to discuss the challenges and come up with a...
-
Over the circle X2 + Y2 (a) What is fY|X(y|x)? (b) What is E[Y|X = x]? otherwise.
-
When a company has a contract involving multiple performance obligations, how must the company recognize revenue?
-
A car travels to the left at a steady speed for a few seconds, then brakes for a stop sign. Draw a complete motion diagram of the car?
-
A goose flies toward a pond. It lands on the water and slides for some distance before it comes to a stop. Draw the motion diagram of the goose, starting shortly before it hits the water and assuming...
-
A goose flies toward a pond. It lands on the water and slides for some distance before it comes to a stop. Draw the motion diagram of the goose, starting shortly before it hits the water and assuming...
-
11. A user forgets to type one of the zeros in one of the steps during creating general ledger reconciliation accounts for accounts receivable, sale revenue, and sales discounts. Then receives error...
-
Required information Skip to question [The following information applies to the questions displayed below.] Drs. Glenn Feltham and David Ambrose began operations of their physical therapy clinic,...
-
On January 1, 2021, the general ledger of Tripley Company included the following account balances: Accounts Debit Credit Cash $286,000 Accounts receivable 76,000 Allowance for uncollectible accounts...

Study smarter with the SolutionInn App


