Predict the major product from each of the following reactions. H O (1) (2) HO+ (3) HCrO4
Question:
Predict the major product from each of the following reactions.
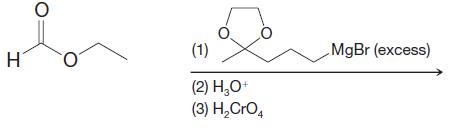
Transcribed Image Text:
H O (1) (2) H₂O+ (3) H₂CrO4 MgBr (excess)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (12 reviews)
For the first reaction the major product is expected to be 2butanol This is because the reactant 2br...View the full answer

Answered By

BillClinton Muguai
I have been a tutor for the past 5 years. I have experience working with students in a variety of subject areas, including computer science, math, science, English, and history. I have also worked with students of all ages, from elementary school to college. In addition to my tutoring experience, I have a degree in education from a top university. This has given me a strong foundation in child development and learning theories, which I use to inform my tutoring practices.
I am patient and adaptable, and I work to create a positive and supportive learning environment for my students. I believe that all students have the ability to succeed, and it is my job to help them find and develop their strengths. I am confident in my ability to tutor students and help them achieve their academic goals.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry
ISBN: 978-1118875766
12th Edition
Authors: T. W. Graham Solomons, Craig B. Fryhle, Scott A. Snyder
Question Posted:
Students also viewed these Sciences questions
-
Predict the major product from each of the following reactions. (a) (b) (c) (d) (e) (f) OH SOC, pyr OH HBr NaNH2 OH OH (1) TsCI, pyr (2) EtSNa Nal, H2SO OH
-
Predict the major organic product from each of the following reaction sequences. (a) (b) (c) (d) (e) (f) (1) MeMgBr (excess) (2) NH,CI, H20 (1) Mg (2) H H (3) HyO (1) PBr3 (2) Mg (3) H,o OH OH (1)...
-
Predict the major organic product from each of the following reactions. (a) (b) (c) (d) (1) CH MgBr (2) HO (1) MgBr (2) NH,CI, H20 MgBr (1 equiv.) O (1) o (2) H,O (1) CH,CHLi (excess) (2) NH,CI, H2O O
-
choess answaer that How many bits are required to store one BCD digit?" 2 8 1 Kilo bits are equal to 1000 bits 1024 bits 1012 bits 1008 bits The function of the encoder is to convert coded...
-
3x > 9 Describe the solution set as an inequality, in interval notation, and on a graph.
-
As I entered the data into an R file for Exercise 12.11 I became a bit concerned about what they meant. Can you suggest a reason for my concern? Exercise 12.11 The data referred to in Exercise 12.10...
-
Your company sells $100,000 of goods and you collect sales tax of 3%. What current liability does the sale create? a. Accounts Payable of $3,000 b. Unearned Revenue of $3,000 c. Sales Revenue of...
-
Following is a partially completed balance sheet for Epsico, Inc., at December 31, 2010, together with comparative data for the year ended December 31, 2009. From the statement of cash flows for the...
-
Tech Manufacturing Corporation reports the following situations in 2020 with respect to its high-tech manufacturing equipment. a.Machine 1 was acquired at a cost of $872,000 in 2017.The machine was...
-
Modify the warehouse location model as suggested in Modeling Issue 2. Specifically, assume that the same four customers have the same annual shipments, but now, there are only two possible warehouse...
-
Show how the diketone at the right could be prepared by a condensation reaction: lion to bolo HO R OEt + + R EtOH
-
Provide a mechanism for the following reaction. NO NO 2 ss-sr NaOCH3 OH NO O=5 S=0 NO
-
Recall the focus of the SANS Institute and the topic areas they cover in their security-focused project management course: Earned value technique (EVT) Leadership and management strategy Project...
-
OHIN ww Decide for what yo, the solution to the following differential equation: y = (sin t)ev, y(0) = 30 exists for all t 0. Next suppose yo = 0, decide the largest T+ (including +00) such that the...
-
Prepare the Statement of Financial Position of Sherwin's Delivery Services using the information of their first month of operation for the month of November 2019. The business had the following...
-
On March 1st, 2018, Firm A decides to enter a semi-annual, 2-year interest rate swap with Bank B, the notional being 1 million USD. The continuously compounded interest rates on March 1st, 2018 are...
-
1 - What is beta? 2 - Stock ABC has a beta of 1.25 and Stock XYZ has a beta of 0.55. Which stock would you buy if you knew the stock market was going to drop by 20% over the next month? Also, briefly...
-
Question 1 information Jerome Yoong plans to market his custom brand of packaged trail mix in Australia's major supermarkets (Coles, Woolworths, Aldi, and Costco), given his current success in the US...
-
Using 30 observations, the following regression output is obtained from estimating the linear probability model y = 0 + 1x + . a. What is the predicted probability when x = 20? b. Is x significant at...
-
How does the organizational structure of an MNC influence its strategy implementation?
-
Which of these stereo isomers has the faster rate of E2elimination? D D H Br Br D.
-
Show the products of this elimination reaction and explain which ismajor: t-BUOH PHCHCH CI + 1-BuO
-
Explain why deuterium is lost in preference to hydrogen in this Hofmann eliminationreaction: *N(CH)3 D. H. + (CH)N + HOD
-
A scuba diver is exploring a shipwreck at a depth of 30 meters in the ocean. The diver's tank contains 5.0 moles of compressed air at a temperature of 25C. What is the pressure of the air in the tank...
-
1. Determine the atmospheric pressure at a location where the barometric reading is 720 mm Hg. 2. A pressure gauge connected to a tank reads 52 psi at a location where the barometric reading is 29.6...
-
Assume that a bond has an annual coupon rate of 6.80 percent, but makes coupon payments on a semi-annual basis, has a par value of $1,000, a yield to maturity of 7.20 percent, and a current price of...

Study smarter with the SolutionInn App


