Which of the following are chiral? H3C H;C H3C CH3 CH3 H3C CH3 CH3 H;C CI H;C
Question:
Which of the following are chiral?
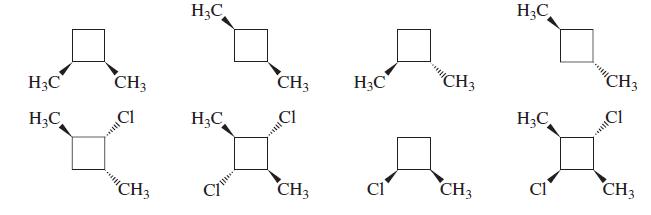
Transcribed Image Text:
H3C H;C H3C CH3 CH3 H3C CH3 "CH3 H;C CI H;C CI H;C CH3 CH3 Cl CH3 CI CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 71% (14 reviews)

Answered By

Salman Ahsan
Did my graduation as well as master's from a reputed central university of India i.e Jamia Millia Islamia in New Delhi. Qualified CSIR-NET (a national level entrance examination for doctorate) in Chemical Sciences, with a national rank of 19. Have been teaching at various institutions from my bachelor's days. Currently working in the field of Ekectroorganic Synthesis at a premier research institute of India i.e. Indian Institute of Science Education and Research Bhopal.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Which of the following objects are chiral? (a) Screwdriver (b) Screw (c) Beanstalk (d) Shoe
-
Which of the following are chiral? (a) 2-Methyl-2,3-hexadiene (b) 4-Methyl-2,3-hexadiene (c) 2,4-Dimethyl-2,3-pentadiene
-
Which of the following objects are chiral and which are achiral? a. Golf club b. Teacup c. Football d. Corkscrew e. Tennis racket f. Shoe g. Portrait h. Pencil
-
When members of your team coordinate how to prepare the progress report, in which direction is the flow of communication?
-
Briefly describe how a magnetic sector mass spectrometer works.
-
Discuss: a. What do you consider a really good example of a celebrity for one of the personality dimensions? Why do you think this person best depicts this dimension? b. Are there any examples that...
-
What conditions are required for valid inferences about the bs in simple linear regression? LO9
-
The Clearinghouse Sweepstakes has just informed you that you have won $1 million. The amount is to be paid out at the rate of $20,000 a year for the next 50 years. With a discount rate of 10 percent,...
-
How does the forward premium or discount relate to interest rate differentials under CIRP?
-
An asset is worth $10,000. The likelihood of an attack is once every two years with an expected impact of 80% damage. The risk acceptance level is $2000. A control that would reduce the risk to $1000...
-
Which of the following compounds has a stereoisomer that is a meso compound? a. 2,4-dibromohexane b. 2,4-dibromopentane c. 2,4-dimethylpentane d. 1,3-dichlorocyclohexane e. 1,4-dichlorocyclohexane f....
-
There are two stable isotopes of nitrogen, 14 N and 15 N. How many protons and neutrons does each isotope have?
-
Show that the process in equation (5.110), \(x_{t}=-\sum_{j=0}^{\infty} \phi_{1}^{-j} w_{t+j}\) where \(\left|\phi_{1} ight| <1\), is stationary. Note an important property: in this representation...
-
Write out the form of the partial fraction decomposition of the function (See Example). Do not determine the numerical values of the coefficients. (If the partial fraction decomposition does not...
-
Listed here are the costs associated with the production of 1,000 drum sets manufactured by TrueBeat. Costs 1. Plastic for casing$16,000 2. Wages of assembly workers$83,000 3. Property taxes on...
-
The diagram shows the instant when a long slender bar of mass 4.8 kg and length 2.9 m is horizontal. At this instant the mass m= 6.2 kg has a vertical velocity of 5.3 m/s. If the pulley has...
-
Holland has been down to the Law Clinic and has returned with several new cases/clients; Wanda and the doorman from the 7-Seas Bar/Grill,Wanda, who has been charged with malicious destruction of a...
-
2) A spring of constant k=1.2 N/cm is being compressed 5-cm by a 200-gram ball. The ball is released on a 10 degree incline. Determine: a) The speed of the ball when it leaves the spring b) The...
-
A table of values of a function f with continuous gradient is given. Find C F dr, where C has parametric equations x = t 2 + 1 y = t 3 + t 0 t 1 y 1 1 6. 4 1 5 8 9 2. 2. 3. 2.
-
Outline a general process applicable to most control situations. Using this, explain how you would develop a system to control home delivery staff at a local pizza shop.
-
Which compound should undergo substitution of the bromine by phenolate anion: 4 - bromopyridine or 3 - bromopyridine ? Explain, and give the structure of the product.
-
Outline a synthesis for each of the following compounds from the indicated starting material and any other reagents. 4-methyl-3-nitropyridine from - lpicoline
-
Predict the predominant product in each of the following reactions. Explain your answer. 3,4-dibromopyridine + NH3, heat (C5H5BrN2)
-
assume that we have only two following risk assets (stock 1&2) in the market. stock 1 - E(r) = 20%, std 20% stock 2- E(r) = 10%, std 20% the correlation coefficient between stock 1 and 2 is 0. and...
-
Flexible manufacturing places new demands on the management accounting information system and how performance is evaluated. In response, a company should a. institute practices that reduce switching...
-
Revenue and expense items and components of other comprehensive income can be reported in the statement of shareholders' equity using: U.S. GAAP. IFRS. Both U.S. GAAP and IFRS. Neither U.S. GAAP nor...

Study smarter with the SolutionInn App


