All of the following acid-base reactions are reactions that we will study in greater detail in the
Question:
(a)
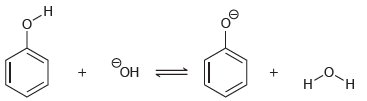
(b)
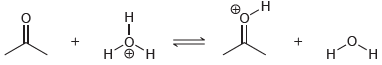
(c)
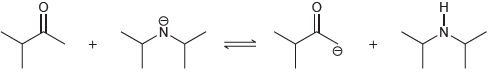
(d)
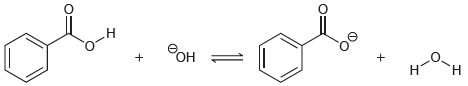
Transcribed Image Text:
Н eон Н H. H.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 73% (15 reviews)
a b c d Aci...View the full answer

Answered By

Antony Sang
I am a research and academic writer whose work is outstanding. I always have my customer's interests at heart. Time is an important factor in our day to day life so I am always time conscious. Plagiarism has never been my thing whatsoever. I give best Research Papers, Computer science and IT papers, Lab reports, Law, programming, Term papers, English and literature, History, Math, Accounting, Business Studies, Finance, Economics, Business Management, Chemistry, Biology, Physics, Anthropology, Sociology, Psychology, Nutrition, Creative Writing, Health Care, Nursing, and Articles.
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Write the products of the following acid-base reactions: (a) CH3OH + H2SO4 ? (b) CH3OH + NaNH2 ? (c) CH3NH3 + C1- +NAOH ?
-
Predict the products of the following acid-base reactions, and predict whether the equilibrium lies to the left or to the right of the equation:
-
Predict the products of the following acid-base reactions, and predict whether the equilibrium lies to the left or to the right of the equation:
-
McGuire Industries prepares budgets to help manage the company. McGuire is budgeting for the fiscal year ended January 31, 2021. During the preceding year ended January 31, 2020, sales totaled $9,200...
-
Obtain an annual report from a publicly traded corporation that is interesting to you. Be sure the company has property and equipment, intangible assets , and long-term debt on its balance sheet....
-
Find the least squares regression line for the points. Use the regression capabilities of a graphing utility to verify your results. Use the graphing utility to plot the points and graph the...
-
Explain various classifications of overheads.
-
American Steel Corp. acquired the following securities in 2011: At the beginning of 2011, American Steel had a zero balance in each of its market adjustment accounts. 1. What entry or entries would...
-
The controller of Hendershot Corporation estimates the amount of materials handling overhead cost that should be allocated to the company's two products using the data that are given below: Wall...
-
During a period of severe inflation, a bond offered a nominal HPR of 80% per year. The inflation rate was 70% per year. a. What was the real HPR on the bond over the year? b. Compare this real HPR to...
-
Identify any formal charges in the following structures: a. b. c. d. -N=N:
-
Each of the following mechanisms contains one or more errorsthat is, the curved arrows may or may not be correct. In each case, identify the errors and then describe what modification would be...
-
On October 1, 2014, Kingsway Broadcasting purchased for $288,000 the copyright to publish the music composed by a local Celtic group. Kingsway expects the music to be sold over the next three years....
-
The transmitted energy expands out into space as it propagates at 3 GHz between the transmitter and the receiver over 30 km distance. Calculate the free space loss using a suitable formula and any...
-
What is the company featured in this episode of Undercover Boss? List 3 good professional activities that the CEO/president learned about their company by going undercover? List areas of the...
-
Assume there is a national lottery in the winning ticket is worth $10 million one winning ticket will be selected if there are 225 million tickets sold. What is the chance that a buyer of one ticket...
-
Description: Reference: Basu Thakur. (2015). PostcolonialTheory and Avatar (pp. 85-150,157-172). Bloomsbury PublishingUSAPre-Peer Paper Review for the Postcolonial Application Paper 1: Collecting...
-
NOT ASKING THE ACTUAL SHEAR STRESS. Please READ! Derive the shear stress distributed equation over the cross-section. Derive the equation and plot. 15 15 30 15 15 120 -90 20 0.5 m 72 kN 20 20 40 40...
-
Solve each system using the elimination method. If a system is inconsistent or has dependent equations, say so. x - 4y = 2 4x 16y=8
-
What is the mode?
-
The 1H NMR peak for the hydroxyl proton of alcohols can be found anywhere from d 0.5 to d 5.4. Explain this variability.
-
(a) Show that replacing each of the CH2 protons by some group Q in the (S) enantio-mer of 2-butanol leads to a pair of diastereomers, as it does for the (R) enantiomer. (b) How many chemically...
-
The 1H NMR study of DMF (N, N-dimethylformamide) results in different spectra according to the temperature of the sample. At room temperature, two signals are observed for the protons of the two...
-
You have just been hired as a new management trainee by Earrings Unlimited, a distributor of earrings to various retail outlets located in shopping malls across the country. In the past, the company...
-
Brief Exercise 10-6 Flint Inc. purchased land, building, and equipment from Laguna Corporation for a cash payment of $327,600. The estimated fair values of the assets are land $62,400, building...
-
"faithful respresentation" is the overriding principle that should be followed in ones prepaparation of IFRS-based financial statement. what is it? explain it fully quoting IAS. how this this...

Study smarter with the SolutionInn App


