Consider the following two compounds. How would you distinguish between them using: a) IR spectroscopy? b) 1
Question:
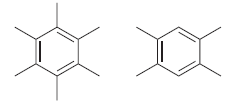
a) IR spectroscopy?
b) 1H NMR spectroscopy?
c) 13C NMR spectroscopy?
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 83% (12 reviews)
a The first compound would lack CH stretching signals just above 3000 cm 1 while the seco...View the full answer

Answered By

Rayan Gilbert
I have been teaching since I started my graduation 3 years ago. As a student, working as Teacher/PA has been tough but made me learn the needs for student and how to help them resolve their problems efficiently. I feel good to be able to help out students because I'm passionate about teaching. My motto for teaching is to convey the knowledge I have to students in a way that makes them understand it without breaking a sweat.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
How would you distinguish between the following compounds using 13 C NMR spectroscopy?
-
How would you distinguish between each pair of compounds using mass spectrometry? a. b. N.
-
How would you distinguish between each pair of compounds using high-resolution mass spectrometry? a. b. .
-
Prepare journal entries to record each of the following sales transactions of TFC Merchandising. TFC uses a perpetual inventory system and the gross method. May 1 9 Sold merchandise for $600, with...
-
What advantages and disadvantages does offering trade credit provide to a small business?
-
A mass-spring chain consists of four masses suspended between two fixed supports. The spring stiffnesses are c1 = 1, c2 = 1/2, c3 = 2/3, c4 = 1/2, c5 = 1. (a) Determine the equilibrium positions of...
-
Determine if \(5,067,322\) is prime or composite.
-
The emergency services coordinator for Clarke County is interested in locating the countys two ambulances to maximize the number of residents that can be reached within four minutes in emergency...
-
If an investment using the value implied by the median EV/EBITDA multiple from the comps is made at the transaction date, what is the implied IRR? Assume the terminal value is the average of the...
-
As of January 1, 2016, Room Designs Inc. had a balance of $9,900 in Cash, $3,500 in Common Stock and $6,400 in Retained Earnings. These were the only accounts with balances in the ledger on January...
-
Consider the equilibrium C 2 H 6 (g) C 2 H 4 (g) + H 2 (g). At 1000.K and a constant total pressure of 1.00 bar, C 2 H 6 (g) is introduced into a reaction vessel. The total pressure is held constant...
-
The following compound cannot be made with either a Friedel-Crafts alkylation or acylation. Explain.
-
For the following mechanism, identify the sequence of arrow-pushing patterns: . :H :H .R R R H20 . jata
-
In its first year, Joe's Start-Up Company had revenues of $125,000 and cost of goods sold of $81,250, which was the only variable cost. Depreciation was $20,000, and cash costs were as follows:...
-
On July 1, 2019, Farm Fresh Industries purchased a specialized delivery truck for $175,600. At the time, Farm Fresh estimated the truck to have a useful life of eight years and a residual value of...
-
Examine and explain the two UNSDGs Goal 6 and Goal 11. In your discussion use at least two different kinds of example projects related to Civil Engineering and Construction Management. Point out the...
-
Links Ltd, a logistics company, is considering buying a smaller competitor in the same industry & operates in the competitor Smartmove Ltd, is a more technologically advanced, but resoutca limited,...
-
A student wishes to use their knowledge of thin lenses to make a simple projector.She wants to create a 9 3 cm tall image from a 2 . 4 cm tall slide. If the screen is 3 . 0 6 maway from the slide,...
-
Heights of women have a bell-shaped distribution with a mean of 161 cm and a standard deviation of 7 cm. Using the empirical rule, what is the approximate percentage of women between a. 154 cm and...
-
You are maintaining a subsidiary ledger account for Police-Training Expenditures for 2013. The following columns are used: Inventory purchases are initially recorded as expenditures. Record the...
-
Draw the structure of each of the following. (Some parts may have more than one correct answer.) (a) A nine-carbon ether that can be prepared by the Williamson synthesis. (b) An ether that would...
-
Draw the structure of each of the following. (Some parts may have more than one correct answer.) (a) A nine-carbon ether that can be prepared by the Williamson synthesis. (b) An ether that would...
-
Give the products of the reaction of 2-ethyl- 2-methyloxirane (or other compound indicated) with each of the following reagents. (a) Water, H3O+ (b) Water, NaOH, heat (c) NA+ CH3O- in CH3OH (d) CH3OH...
-
Could you show the steps with detailed explanations? Thanks!. Determine whether each of the following integer sequences are graphic or not. For any that are graphic, give a graph that realizes it...
-
To begin, you will take the rules of your game that you provided in your Deliverable 1 Design Document and turn them into fully developed use cases for your game. deliverable 1: Project Object...
-
Cars (35 points) Implement a class Car, which contains the fields (5 points): make, e.g. Ford, Subaru, Toyota ... model, e.g., Escape, Outback, Camry ... year MPG miles per gallon milesDriven, the...

Study smarter with the SolutionInn App


