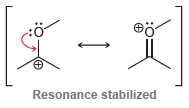
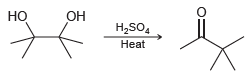
Determine whether the pinacol rearrangement, shown in the previous problem, is a reduction, an oxidation, or neither.
Question:
Transcribed Image Text:
Resonance stabilized ОН Но H,SO, Нeat
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (10 reviews)
One carbon atom is oxidized from an oxidation state of 1 to ...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The outline of the part in the previous problem is to be profile milled using a 30 mm diameter end mill with four teeth. The part is 15 mm thick. Cutting speed = 150 mm/min and feed = 0.085 mm/tooth....
-
The density of the apples in the previous problem is 0.8 g/cm3 and the density of the oranges is 0.95 g/cm3. What is the total volume if you add the apples to the oranges? Compute the answers by...
-
The reaction in the previous problem is an equilibrium process. Draw a mechanism of the reverse process. That is, draw a mechanism showing conversion of the conjugated, cyclic enone into the acyclic...
-
Vito Co.'s next dividend is expected to be $4.50. Dividend growth is estimated at 20%, 15%, 8% for the following three years, and then stabilize to 2%. How much are you willing to pay to buy one...
-
What is the accounting equation? Why is this important to the practice of accounting? Please provide an example of how the accounting equation works with recording transactions within a business....
-
Find a point on the ellipsoid x + 4y + z = 9 where the tangent plane is perpendicular to the line with parametric equations
-
1 Why did the IT expert feel demotivated?
-
The following on-boarding process for new employees has two stages. In the first, employees fill out several government forms and necessary visas for immigration. In the second the employees attend...
-
For each statement, indicate the accounting term described. \ table [ [ Costs that remain unchanged despite changes in sales volume. ] , [ The reduction in unit cost achieved from a higher level of...
-
Determine the maturity date and compute Interest for each note. Contract Date Interest Note 1. 2. Principal $17,000 22,000 15,000 Period of Nete (Tern) 60 days 90 days 45 days Rate March B 6% May 22...
-
One liter of fully oxygenated blood can carry 0.18 liters of O 2 measured at T = 298 K and P = 1.00 atm. Calculate the number of moles of O 2 carried per liter of blood. Hemoglobin, the oxygen...
-
Determine whether the pinacol rearrangement, shown in the previous problem, is a reduction, an oxidation, or neither. Resonance stabilized H,SO, eat
-
GianAuto Corporation manufactures automobiles, vans, and trucks. Among the various GianAuto plants around the United States is the Denver cover plant, where vinyl covers and upholstery fabric are...
-
Q10: Region ( experienced compressive stresses and has a than the rest of the bracket. Region ( ) experienced tension stresses and has a of the bracket. Deep Drawing and Stretch Forming width (into...
-
A sample of 1500 computer chips revealed that 32% of the chips do not fail in the first 1000 hours of their use. The company\'s promotional literature claimed that above 29% do not fail in the first...
-
The 75 lb block is released from rest 5 ft above the plate. Determine the compression of each spring when the block momentarily comes to rest after striking the plate. Neglect the mass of the plate....
-
Indiana Soy Products (OSP) buys soybeans and processes them into other soy products. Each ton of soybeans that OSP purchases for $250 can be converted for an additional $180 into 675 lbs of soy meal...
-
The 2025 Annual Report of Splish International contains the following informatio (in millions) June 29, 2025 June 27, 2024 Total assets $1,545 $1,502 Total liabilities 989 1,060 Net sales 2,800 2.971...
-
Louisiana Products Co. had the following positive cash flows during the current year: received cash from customers of $750,000; received bank loans of $35,000; and received cash from the sale of...
-
Nitrogen monoxide reacts with hydrogen as follows: 2NO(g)+ H2(g) N2O(g) + H2O(g) The rate law is [H2]/ t = k[NO]2[H2], where k is 1.10 107 L2/(mol2s) at 826oC. A vessel contains NO and H2 at...
-
Diethylstilbestrol (DES) has estrogenic activity even though it is structurally unrelated to steroids. Once used as an additive in animal feed, DES has been implicated as a causative agent in several...
-
Propose a synthesis of diethylstilbestrol (Problem 27.44) from phenol and any other organic compound required.
-
What products would you expect from reaction of estradiol (Problem 27.44) with the following reagents? (a) NaH, then CH3I (b) CH3COCI, pyridine (c) Br2, FeBr3 (d) Pyridinium chlorochromate in CH2Cl2
-
Your company produces a health magazine. Its sales data for 1 - year subscriptions are as follows: Year of Operation Subscriptions Sold % Expired at Year End 2 0 2 0 $ 3 0 0 , 0 0 0 5 0 2 0 2 1 $ 6 4...
-
The adjusted trial balance for Tybalt Construction on December 3 1 of the current year follows. TYBALT CONSTRUCTION Adjusted Trial Balance December 3 1 Number Account Title Debit Credit 1 0 1 Cash $...
-
( US$ millions ) 1 2 / 3 1 / 2 0 1 4 1 2 / 3 1 / 2 0 1 3 1 2 / 3 1 / 2 0 1 2 1 2 / 3 1 / 2 0 1 1 Net income $ 1 4 , 4 3 1 $ 1 2 , 8 5 5 $ 1 0 , 7 7 3 $ 9 , 7 7 2 Depreciation 3 , 5 4 4 2 , 7 0 9 1 ,...

Study smarter with the SolutionInn App


