Draw bond-line structures for vitamin A and vitamin C: . 1 -
Question:
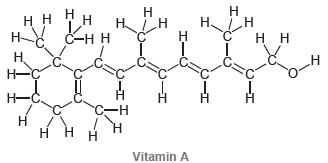
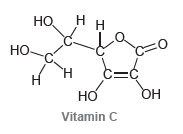
Н Н. н Н н 1н нн н С-нн Нн нн Н Н ннн Н Vitamin A но. нн Но. C=Ć он н но Vitamin C
Step by Step Answer:
Vitami...View the full answer



Related Video
Lemon juice preserves apples by slowing down the oxidation process. Oxidation is a chemical reaction that occurs when oxygen reacts with certain substances, such as apples. When an apple is cut or bitten, oxygen is exposed to the inside of the apple and causes enzymes in the apple to turn brown, which is an indication of oxidation. The browning process is caused by the production of polyphenol oxidase (PPO) enzymes that convert phenolic compounds into quinones, which then polymerize to form the brown pigments. One of the compounds present in lemon juice is ascorbic acid (vitamin C), which is a natural antioxidant. Antioxidants work by neutralizing the free radicals that cause oxidation. When lemon juice is applied to apples, the ascorbic acid in the lemon juice reacts with the PPO enzymes and slows down the browning process. You can do an experiment by cutting apples into small pieces, leaving one apple piece in contact with air and the others covered with lemon juice and compare the browning process. This will help to understand the antioxidation process in fruits.
Students also viewed these Sciences questions
-
Draw bond-line structures for all constitutional isomers with molecular formula C 4 H 9 Cl.
-
Draw bond-line structures for all constitutional isomers of the following compound: CH 3 CH 2 CH(CH 3 ) 2
-
Draw bond-line structures for all constitutional isomers of C 4 H 10 ?
-
Darrel & Co. makes electronic components. Chris Darrel, the president, recently instructed Vice President Jim Bruegger to develop a total quality control program. If we dont at least match the...
-
On March 1, Jordan Company borrows $150,000 from Ottawa State Bank by signing a 6-month, 8%, interest-bearing note. Instructions prepare the necessary entries below associated with the note payable...
-
There are three carbon-oxygen bonds in methyl acetate. a. What are their relative lengths? b. What are the relative infrared (IR) stretching frequencies of these bonds?
-
Smith and Smith & Associates is contemplating the purchase of equipment that would cost \($196,600\). Currently, the company rents similar equipment for \($45,076\) per year. The proposed new...
-
Kragan Clothing Company manufactures its own designed and labeled sports attire and sells its products through catalog sales and retail outlets. While Kragan has for years used activity-based costing...
-
Question 8 The December 2022 S&P 500 cash index is 3980 while the S&P 500 futures index is 4000 and the contract value of each index point on the futures is $100. You are convinced the cash...
-
Trumball Catering served 4,000 meals last month. Trumball recorded the following costs with those meals: Required Unit variable costs and total fixed costs are expected to remain unchanged next...
-
Draw bond-line structures for all constitutional isomers of C 5 H 12 ?
-
How many lone pairs are found in the structure of vitamin C?
-
Consider the following experimenta card is chosen randomly from a 52-card deck, observed, and not replaced. A second card is drawn and observed. Consider the following events: A = The first card is...
-
Solve the differential equation: dy +2y=3e* dx Find the general solution to the second-order linear homogeneous differential equation: y" - 6y' + 9y=0
-
Russia is exporting mostly commodities. Below find Russia's terms of trade (TOT) during 2012-2019. Russia import/GDP at constant national prices is 0.2 1. What explains the deterioration of Russia's...
-
The voltage and current at the terminals of this circuit are zero for t <0. Fort20 v= 40 e 1,500 Volts i=4e-1,500 Amps a) Find the power absorbed/delivered by the circuit element at t= 500 s (micro...
-
1.The RN is performing an assessment on a patient being admitted for back pain. The nurse interprets which of the patients statements as a minor detail? a.The patient has not been able to void in 12...
-
The Horse Place is a retailer that sells horse tack (saddles, saddle pads, bridles, reins, etc.) and supplies (grooming products, vitamin supplements, leg wraps, etc.). The Horse Place has 15 stores...
-
Listed below are measured loads (in lb) on the left femur of crash test dummies used in the same cars listed in the Chapter Problem. Use a 0.05 significance level to test the null hypothesis that the...
-
The polar coordinates of a point are given. Find the rectangular coordinates of the point. (-1, - /3)
-
Compounds A, B, and C are isomers with the formula C5H11Br. Their broadband proton-decoupled 13C NMR spectra are given in Fig. 9.32. Information from the DEPT 13C NMR spectra is given near each peak....
-
In contrast to 2-methylbutane and neopentane, the mass spectrum of 3-methylpentane (not given) has a peak of very low relative abundance at - 15. It has a peak of very high relative abundance at -...
-
Propose structures and fragmentation mechanisms corresponding to ions with m/z 57 and 41 in the mass spectrum of 4-methyl-1-hexene. onization (loss of a electron) mlz 57 m/z 41
-
23 BUSM 1260-22 Applied Business Mathematics/Calculators ezto.mheducation.com M Question 1- Chapter 14 Homework by Sunday, 11/12 - Connect 4 Homework by Sunday, 11/12 ok nt rences Complete the...
-
ABC Bank has assets of $120 and a leverage ratio of 3. It earns 5% on its assets but pays 4% on its liabilities. What is ABC Bank's profit per unit of capital invested? X (round to the nearest...
-
Exercise2 (10 points) Background readings: Chapter 2 For the below graph, and using the data information in it, create a data table in order to organize data for processing by computers or for...

Study smarter with the SolutionInn App


