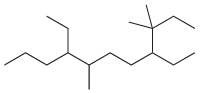
For each of the following compounds, identify all groups that would be considered substituents, and then indicate
Question:
(a)
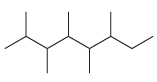
(b)
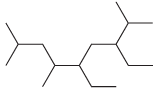
(c)
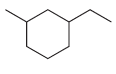
(d)

(e)
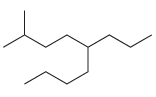
(f)
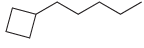
(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 72% (18 reviews)
a b c d ...View the full answer

Answered By

ANDREW KIPRUTO
Academic Writing Expert
I have over 7 years of research and application experience. I am trained and licensed to provide expertise in IT information, computer sciences related topics and other units like chemistry, Business, law, biology, biochemistry, and genetics. I'm a network and IT admin with +8 years of experience in all kind of environments.
I can help you in the following areas:
Networking
- Ethernet, Wireless Airmax and 802.11, fiber networks on GPON/GEPON and WDM
- Protocols and IP Services: VLANs, LACP, ACLs, VPNs, OSPF, BGP, RADIUS, PPPoE, DNS, Proxies, SNMP
- Vendors: MikroTik, Ubiquiti, Cisco, Juniper, HP, Dell, DrayTek, SMC, Zyxel, Furukawa Electric, and many more
- Monitoring Systems: PRTG, Zabbix, Whatsup Gold, TheDude, RRDtoo
Always available for new projects! Contact me for any inquiries
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Name the following compounds: a. b. c. d. e. f. Br
-
Name the following compounds: a. b. c. d. e. f. CH3 CH3 CH2CH3 CH3 HN CH3 CH3 CH3 CH-CH3
-
Name the following compounds: (a) (b) (c) (d) (e) (f) (g) (h) CH,CH,C CH OH CH CH CH NO COOH Br OCH(CH)2 OH NO NO CH,OCH,CH,
-
In Exercises verify the identity. coshx = 1 + cosh 2x 2
-
Explain the organizational archetypes as described by Mintzberg and discuss how organizational structure influences innovation. Summarize Mintzberg's work with archetypes. What key features and...
-
Find the expected values and the standard deviations (by inspection) of the normal random variables with the density functions given in Exercises. 1 3V2 e-(1/18)x
-
Trends. Which of these series of data do you expect to show a clear trend? Will the trend be upward or downward? (All data are recorded annually.) (a) The percentage of students entering a university...
-
What social and/or cultural factors played a role in this consumer purchasing the smartphone and data plan? Which ones do you think were most important and why?
-
Coronado Company acquired a plant asset at the beginning of Year 1. The asset has an estimated service life of 5 years. An employee has prepared depreciation schedules for this asset using three...
-
There is a lottery with n coupons and n people take part in it. Each person picks exactly one coupon. Coupons are numbered consecutively from 1 to n, n being the maximum ticket number. The winner of...
-
Explain the relationship between the terms exact differential and state function.
-
In the experiment shown in Figure 2.4b, the weight drops in a very short time. How will the temperature of the water change with time? Figure 2.4b Electrical generator Mass Heating - coil (b)
-
Divide: a. x 3 + x + 10 by (x + 2) b. 2x 3 17x + 3 by (x + 3) c. 3x 3 + 50x 8 by (x 4)
-
Please help. I would really appreciate it. Question 1. Polly owns an electric power plant in the city of Newtown. The market price of electricity in Newtown is $1.00 per kilowatt hour (kwh). Polly's...
-
(Appendix 3A) Jenson Manufacturing is developing cost formula for future planning and cost control. Utilities is one of the mixed costs associated with production. The cost analyst has suggested that...
-
A company has the following trial balance as at 31 December 2015: TRIAL BALANCE AS AT 31 DEC 2015 Dr Cr Sales Revenue 125 000 Purchases 78 000 Carriage 4 000 Electricity and rent 5 100 Administrative...
-
What gets printed to the screen by the following segment of code? String str1 = "hello"; String str2 = "world"; if (!strl.equals(str2)) { System.out.println(str1+" "+str2); } else { }...
-
a) Consider the following financial data (in millions of dollars) for Costello Laboratories over the period of 2014-2018: Year Sales Net income Total assets Common equity 2014 $3,800 $500 $3,900...
-
Two angles of a triangle have the same measure. The measure of the third angle is 4 less than twice the measure of each of the equal angles. Find the measures of the three angles. x (2x-4) ot
-
In the current year, the City of Omaha donates land worth $500,000 to Ace Corporation to induce it to locate in Omaha and create an estimated 2,000 jobs for its citizens. a. How much income, if any,...
-
Assign oxidation states to each carbon of ethanol, acetaldehyde, and acetic acid. [Ol Ethanol Acetaldehyde Acetic acid
-
Provide a retrosynthetic analysis and synthesis for each of the following compounds. Permitted starting materials are phenylmagnesium bromide, oxirane, formaldehyde, and alcohols or esters of four...
-
What products would you expect from the reaction of ethylmagnesium bromide (CH3 CH2MgBr) with each of the following reagents? (a) H2O (b) D2O (c) (d) (e) (f) (g) then H O "H Ph" PhPh, then NH4CI, HO...
-
You are the digital marketing director for High West fashions, a regional clothing company that specializes in custom t-shirts. Your company has decided to launch an online advertising campaign that...
-
In-the-money put options will automatically get exercised at the expiration. True OR False
-
Which of the following examples of business-use property is NOT eligible for Section 1231 treatment when sold at a gain? * Sale of land held for three years. Net gain from a casualty gain on a dump...

Study smarter with the SolutionInn App


