For each of the following compounds, identify the expected chemical shift for each type of proton: (a)
Question:
(a)
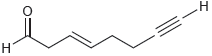
(b)
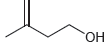
(c)
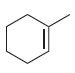
(d)
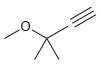
Transcribed Image Text:
н н ОН
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 85% (14 reviews)
a b c d 10 pp...View the full answer

Answered By

Mario Alvarez
I teach Statistics and Probability for students of my university ( Univerisity Centroamerican Jose Simeon Canas) in my free time and when students ask for me, I prepare and teach students that are in courses of Statistics and Probability. Also I teach students of the University Francisco Gavidia and Universidad of El Salvador that need help in some topics about Statistics, Probability, Math, Calculus. I love teaching Statistics and Probability! Why me?
** I have experience in Statistics and Probability topics for middle school, high school and university.
** I always want to share my knowledge with my students and have a great relationship with them.
** I have experience working with students online.
** I am very patient with my students and highly committed with them
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Draw both chair conformations for each of the following compounds. In each case, identify the more stable chair conformation: (a) Methylcyclohexane (b) Trans-1,2-Diisopropylcyclohexane (c)...
-
For each of the following compounds, identify any polar covalent bonds by drawing δ+ and δ- symbols in the appropriate locations. a. b. c. d. e. f. -- :F: - : CI
-
For each of the following compounds, identify whether each C R C system is cumulated, conjugated, or isolated: (a) (b) (c) (d) HO HO- cis-Aconitic acid Plays a role in the citric acid cycle
-
Swish Designs specialises in designing commercial office space in Auckland. The CEO, Ralph Polo has reviewed the financial results and has noticed that operating profits were below budget. He also...
-
Explain the role of inflationary expectations, economic contraction, and Federal Reserve Policy in producing the record low interest rates observed at the end of 2007 and the beginning of 2008.
-
Reginald Smith, an employee of UR Safe Ltd, a security company, broke into a branch of a bank that was a customer of UR Safe Ltd. Smith did so when he was not on shift with UR Safe Ltd, and by using...
-
The cash book of Nagarjuna Restaurant shows8364 as the balance at bank as on 31 December 2002. But you find that this does not agree with the balance as per the bank passbook. On scrutiny, you find...
-
Read Appendix 9-1. Referring to Exhibit 9-14 and related notes, assume instead that Toyozas inventories were costed using the FIFO method and that Lincoln Enterprises employed the LIFO method....
-
Exercise 7.25 Sunland Products manufactures two component parts: A340 and A360. AJ40 components are being introduced currently, and A160 parts have been in production for several years. For the...
-
A 13.6-kg block is supported by the spring arrangement shown. If the block is moved from its equilibrium 44 mm vertically downward and released, determine (a) The period and frequency of the...
-
What data are necessary to construct a financial break-even chart?
-
A compound with molecular formula C 5 H 10 O 2 has the following NMR spectrum. Determine the number of protons giving rise to each signal. Proton NMR lle 2.0 1.5 0.5 ppm 4.5 4.0 33.2 3.5 3.0 2.5 1.0...
-
Give the electron configurations for the following ions: Fe 2+ , Al 3+ , Cu + , Ba 2+ , Br - , and O 2- .
-
Woodland Wearables produces two models of smartwatches, the Basic and the Flash. The watches have the following characteristics:Basic Flash Selling price per watch$ is 270$ 460 Variable cost per...
-
Based on the information provided and recognizing the value of coordinating across its portfolio of businesses, how should LendingTree manage these newer businesses? * as more integrated units * as...
-
Trust Fund Worksheet Background An inter vivos trust was created by Isaac Posney. Isaac owned a large department store in Juggins, Utah. Adjacent to the store, Isaac also owned a tract of land that...
-
A popular theory is that presidential candidates have an advantage if they are taller than their main opponents. Listed are heights (in centimeters) of randomly selected presidents along with the...
-
Gracia Enterprises operates across five industries. Task 1 : After reviewing the information provided, determine which of the five operating segments are reportable based on the revenue test, asset...
-
Find f -1 (x). f(x) = -2x + 10
-
Which of the companies has the lowest accounts receivable turnover in the year 20X2? a. Company A. b. Company B. c. Company C. d. CompanyD. 20X1 20X2 Credit Sales Average Receivables Balance $1.0...
-
Draw the most stable conformation of 1, 4-dichiorobutane, using wedges and dashes to represent bonds coming out of the paper and going behind the paper, respectively.
-
Malic acid, C4H6O5, has been isolated from apples. Because this compound reacts with 2 molar equivalents of base, it is a dicarboxylic acid. (a) Draw at least five possible structures. (b) If malic...
-
Formaldehyde, H2C = O, is known to all biologists because of its usefulness as a tissue preservative. When pure, formaldehyde trimerizes to give trioxane, C3H6O3, which, surprisingly enough, has no...
-
A company manufactures lawnmowers. Compute the total amount of period costs from thr following costs.
-
TestAnswerSavedHelp opens in a new windowSave & ExitSubmit Item 1 7 1 0 points Time Remaining 1 hour 2 0 minutes 1 8 seconds 0 1 : 2 0 : 1 8 Item 1 7 Time Remaining 1 hour 2 0 minutes 1 8 seconds 0 1...
-
Use the following information for the Problems below. (Algo) [The following information applies to the questions displayed below.] Lansing Company's current-year income statement and selected balance...

Study smarter with the SolutionInn App


