For each of the following pairs of compounds, identify the higher boiling compound and justify your choice:
Question:
a.
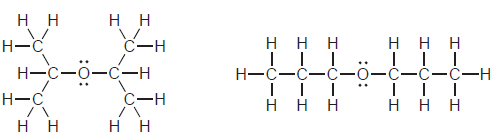
b.
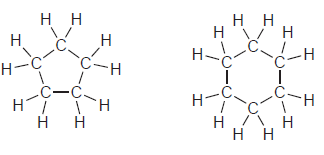
c.
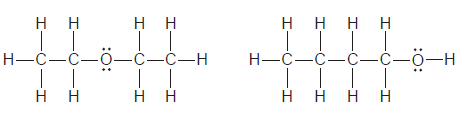
d.
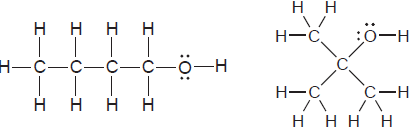
Transcribed Image Text:
нн нн Н-С ннн ннн С—Н Н-с—о—с—н Н—с—с—с—о-с—с—с—н С—н Н-С ннн ннн нн нн нн н н нн Н Н н- -н н-с" c-C- -н н н н- TH н., н нн нн
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (10 reviews)
a The latter because it is less b...View the full answer

Answered By

Parvesh Kumar
I am an experienced Mathematics and Statistics tutor with 10 years of experience teaching students and working professionals. I love teaching students who are passionate to learn subjects or wants to understand any mathematics and statistics concept at graduation or master’s level. I have worked with thousands of students in my teaching career. I have helped students deal with difficult topics and subjects like Calculus, Algebra, Discrete Mathematics, Complex analysis, Graph theory, Hypothesis testing, Probability, Statistical Inference and more. After learning from me, students have found Mathematics and Statistics not dull but a fun subject. I can handle almost all curriculum of mathematics. I did B.Sc (mathematics), M.Sc (mathematics), M.Tech (IT) and am also Gate (CS) qualified. I have worked in various college and school and also provided online tutoring to American and Canadian students. I look forward to discussing with you and make learning a meaningful and purposeful
5.00+
4+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
For each of the following pairs of compounds, give one absorption band that could be used to distinguish between them: a. b. c. d. e. f. CH3CH2CH2CH and CH3CH2OCH3 CH CH2COCH and CH3CH2COH CH3CH2COH...
-
For each of the following pairs of compounds, identify one IR absorption band that could be used to distinguish between them: a. b. c. d. e. f. g. h. i. cis-2-butene and trans-2-butene j. CH3CH2CH2OH...
-
For each of the following pairs of compounds, indicate the compound that you would expect to be a more potent inhibitor of dihydrofolate reductase: a. b. NH NH CI of CH3 CH3 NH2 CH3 or H.NNCHCHCHCH,...
-
Ari Goldstein issued $300,000 of 11%, five-year bonds payable on January 1, 2024. The market interest rate at the date of issuance was 10%, and the bonds pay interest semiannually. Requirements 1....
-
Bob Mark is the executor of his parent's estate that is being divided equally among his two brothers, Tom and Mike, and himself. The estate consist of cash and marketable securities worth $4.2...
-
Locate the numbers z1 + z2 and z1 z2 vectorially when (a) z1 = 2i, z2 = 2/3 - I; (b) z1 = (-3, 1), z2 = (3,0); (c) z1 = (-3, 1) z2 = (1, 4); (d) z1 = x1 + iy1, z2 = x1 - iy1.
-
Can credit derivatives be based on options?
-
Flores Chemical Co. recorded costs for the month of $18,900 for materials, $44,100 for labor, and $26,250 for factory overhead. There was no beginning work in process, 8,000 units were finished, and...
-
Consolidated Croissant Company, Inc. affixes a symbol on all of its croissant packages that features three interlocking C's forming the outline of a croissant. A competitor, the Best Biscuits Bakery,...
-
Dr. Zhivgo Diagnostics Corp. income statements for 20X1 are as follows: Sales $2,790,000 Cost of goods sold 1,790,000 Gross profit 1,000,000 Selling and administrative expense 302,000 Operating...
-
Why is it reasonable to write dH C P dT + VdP for a liquid or solid sample?
-
Refer to Figure 1.10 and explain why (U/V) T is generally small for a real gas. Figure 1.10 Ideal gas Real gas Tranlaition rv-0 (4)A
-
Why are fixed exchange rates a problem for any nation coming under intense speculative international pressure?
-
With three 470 resistors as load, use the other channel of the oscilloscope to observe and note the waveform of the source signal Vs. Compare the part of the cycle that is distorted with the part of...
-
object A object B Type Cup Earth gravitational Cup Table electrostatic Table Earth gravitational Table Earth electrostatic First, identify the interacting bodies, object A and object B. Secondly,...
-
1. You are holding a mass of B kg on top of the 10.0 m height building. a. What is the potential energy of the object with respect to the ground. b. Now you decide to drop the object to the ground,...
-
The "over-the-air" television channel 9 operates between frequencies 186 MHz and 192 MHz, with the video and audio using different parts of this frequency range. To watch (and listen) to the channel...
-
Kp. Show the derived Maxwell-Boltzmann gas partition function a. Assuming the classical without Gibbs correction, b. With classical assumptions but taking Gibbs correction into account, c. By taking...
-
Go to www.charitynavigator.org and scroll down until you find Rating Methodology. Read the information that is provided and write a short memo to explain how this organization rates charities.
-
Transform the while loop from the previous exercise into an equivalent for loop (make sure it produces the same output).
-
Write reaction conditions and the product from Swern oxidation of the following compounds. (a) (b) Myrtenol Chrysanthemyl alcohol
-
Predict the products of the following reactions. (a) (b) (1) EtMgBr (excess) (1) EMgBr (excess) HOEt 2) NH.CI, H2O
-
Predict the organic product from each of the following reduction reactions. (a) (b) (c) OH NaBH (1) LiAIH (2) aq. H2SO NaBH
-
Sandhill Company had the following transactions pertaining to debt investments. 2022 Jan. 1 Purchased 81,7 %, $1,000 Wildhorse Company bonds for $81.000 cash. Interest is payable annually on January...
-
Attempt in Progress Sunland Painting Service has budgeted the following time and material for 2022: BUDGETED COSTS FOR 2022 Time Charges Material Charges Painters' wages and benefits $41.760 Service...
-
At the beginning of 2024, Taylor Corporation had the following stockholders' equity balances in its general ledger Common Stock, $10 Par Value Paid-In Capital in Excess of Par: Common $2,500,000...

Study smarter with the SolutionInn App


