For each pair of compounds below, identify which compound is more acidic and explain your choice: (a)
Question:
(a)
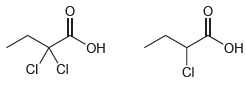
(b)
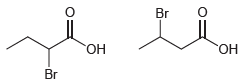
Transcribed Image Text:
ОН "он Но. ČI Br ОН он Он Br
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 63% (11 reviews)
a The compound with two chlorine atoms is more acidic because of the electron with drawi...View the full answer

Answered By

Projeet Mohanty
Growing up I had difficulties learning physics as i did not get the necessary help. Keeping that in mind I have made myself eligible enough to help people with physics. I teach students in class and make the topic interesting by providing relevant examples. Here i would like to help students with their questions and try to provide the best possible answers.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
For each pair of compounds below, determine whether they are identical compounds, constitutional isomers, stereoisomers, or different conformations of the same compound: (a) (b) (c) (d) (e) (f) (g)...
-
For each pair of compounds below, identify the more acidic compound: (a) (b) (c) (d) (e) (f) (g) (h) SH
-
For each pair of compounds below, identify the more acidic compound: (a) (b) (c) (d) (e) (f) (b) C (d) H. -S -3- (f) X H' `H.
-
In Exercises approximate the definite integral using the Trapezoidal Rule and Simpson's Rule with n = 4. Compare these results with the approximation of the integral using a graphing utility. 3.1 J3...
-
1) What is the best way to handle manufacturing overhead costs in order to get the most timely job cost information? A. The company should apply overhead using an estimated rate throughout the year....
-
The temperature at each point on the sphere Let T(x, y, z) = 100 + x + y represent x + y + z = 50. Find the maximum temperature on the curve formed by the intersection of the sphere and the plane x -...
-
Find the margin of error. Should the commissioner of baseball punish Major League Baseball players named in the Mitchell report on steroid use in baseball for having used steroids? A poll of 413...
-
American Food Services, Inc. leased a packaging machine from Barton and Barton Corporation. Barton and Barton completed construction of the machine on January 1, 2011. The lease agreement for the $4...
-
Predetermined Factory Overhead Rate Poehling Medical Center has a single operating room that is used by local physicians to perform surgical procedures. The cost of using the operating room is...
-
Next week, Super Discount Airlines has a flight from New York to Los Angeles that will be booked to capacity. The airline knows from past history that an average of 25 customers ( with a standard...
-
Identify the most acidic proton in each of the following compounds and explain your choice: (a) (b) 0-H F3C 0-H CI. CI
-
Consider the structure of 2,3-dichloropropanoic acid: This compound has many constitutional isomers. (a) Draw a constitutional isomer that is slightly more acidic and explain your choice. (b) Draw a...
-
What is a degenerate system?
-
A. Use the following information to answer the six questions below. Variable Manufacturing Cost Per Unit20 Variable selling cost per unit25 Selling Price per unit100 Fixed Manufacturing cost per unit...
-
The team has been charged with reviewing quarterly results for the LusterLast moisturizing shampoo, called SatinSmooth. The product is new to the line and is sold mostly in drugstores and grocery...
-
Problem 4 (25 pts.) Consider the function f(x, y) = xy y +2. (i) (5 pts) Find the gradient of f (ii) (10 pts) Find the directional derivative of f at the point (1,2) in the direction of the vector...
-
PROBLEM 4. (15 points) a) Determine the range of charged particles emitted from Phosphorus-32 in iron. (5 points) b) Determine the necessary thickness of an iron plate to attenuate the flux of...
-
(b) In the case of no losses, Moody (1965) recommends the following equation for calculating the mass flow rate of wet steam (ie. two-phase water) through the constriction =A 2(h-h) Variable and...
-
Solve each system using the substitution method. If a system is inconsistent or has dependent equations, say so. 3x + 2y = 6 4x + y = 3
-
According to a recent survey, 40% of millennials (those born in the 1980s or 1990s) view themselves more as spenders than savers. The survey also reveals that 75% of millennials view social...
-
Epoxides can be synthesized by treating halohydrins with aqueous base. Propose a mecha-nism for reactions (a) and (b), and explain why no epoxide formation is observed in (c). (a) (b) (c) CI NaOH H20...
-
Write structures for products A, B, C, and D, showing stereochemistry. (Hint: B and D are stereoisomers.) Ko Br OH (H2) TsCl, pyr OH K CO3
-
Propose mechanisms for the following reactions. (a) (b) cat. H.SO Br C3 cat. H.,SO OH
-
Slow Roll Drum Co. is evaluating the extension of credit to a new group of customers. Although these customers will provide $198,000 in additional credit sales, 13 percent are likely to be...
-
Wendell's Donut Shoppe is investigating the purchase of a new $39,600 conut-making machine. The new machine would permit the company to reduce the amount of part-time help needed, at a cost savings...
-
1.Discuss the challenges faced with Valuing Stocks and Bonds. As part of this discussion, how will the selected item be implemented in an organization and its significance? 2. Discuss how Valuing...

Study smarter with the SolutionInn App


