How many stereo-isomers do you expect for the following compound? Draw all of the stereo isomers. ,
Question:
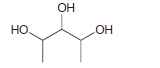
Transcribed Image Text:
ОН но, ОН
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 58% (12 reviews)
There are onl...View the full answer

Answered By

Muqadas Javed
I am a mentor by profession since seven years. I have been teaching on online forums and in universities. Teaching is my passion therefore i always try to find simple solution for complicated problems or task grasp them so that students can easily grasp them.I will provide you very detailed and self explanatory answers and that will help you to get good grade. I have two slogans: quality solution and on time delivery.
4.60+
24+ Reviews
144+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
How many chirality centers are present in estradiol how many stereo isomers does estradiol have?
-
Draw the NMR spectra you expect for the following compounds. (a) (b) (c) (d) C(CH33 CH3O CH3 - Cl C-OCH CH - CH3
-
How many stereo isomers exist for this compound? Assign the relative stabilities of each. Is the methyl group axial or equatorial in the more stable conformer of the least stable stereo isomer? CH3 Ph
-
Which one of the following results from the latest decision round are least important in providing guidance to company managers in making their strategic moves and decisions to improve their...
-
What important events have contributed to the need for ergonomics?
-
Two 8kg blocks are attached to a cord that passes over two fixed drums. If 3 = 0.3 at the drums, determine the equilibrium angle the cord makes with the horizontal when a vertical force p = 200N is...
-
Beyond simply increasing revenue, what advantages might a new business benefit from thanks to early international exposure and growth? L01
-
During 2017, the following transactions were recorded by the Port Hudson Community Hospital, a private sector not-for-profit institution. 1. Gross charges for patient services, all charged to Patient...
-
I need my work to be checked please !! The total debits for all transcations needs to be at $188,656,357 but I'm at $188,653,447. Please check to see if I have the correct journal entries and...
-
Camp and Fevurly Financial Planners have forecasted revenues for the first six months of 2012, as shown in the following table. Month..................... Revenue.....................
-
List 3 variables that you will control.
-
Identify the factors and their levels in the following research designs. a. Men and womens enjoyment of two different sporting events, Sport 1 and Sport 2, are compared using a 20-point enjoyment...
-
A 2.0 kg object is moving to the right with a speed of 1.0 m/s when it experiences the force shown in FIGURE EX11.9. What are the objects speed and direction after the force ends? F, (N) 8. t (s)...
-
If f ( x ) = ( 1 3 - In ( x ) ) ^ 8 , determine f ' ( 1 ) .
-
1. ThestocksAandBhavethefollowingdistributionsofreturns. A B Probability State1 3 4 0.2 State2 5 2 0.3 State3 4 8 0.2 State4 6 5 0.1 State5 6 1 0.2 2....
-
Define nested designs. Explain why the nested designs are important.
-
3 x y 3 + x y = l n ( x ) solve for d y d x
-
Let ln ( xy ) + y ^ 8 = x ^ 7 + 2 . Find dy / dx .
-
If possible, solve the nonlinear system of equations. 2x - y = 0 2xy = 4
-
Discrete sample spaces: suppose there are N cable cars in San Francisco, numbered sequentially from 1 to N. You see a cable car at random; it is numbered 203. You wish to estimate N. (See Goodman,...
-
Explain why the axial strain energies for the C N group and the C CH group are much smaller than that for the CH 3 group.
-
Draw the chair conformations of 1, 1, 3trimethyl cyclohexane. Which conformation is more stable? Why is it not possible on the basis of the material in this chapter to determine the exact energy...
-
Draw the cis and trans stereo isomers of this compound and explain their relative stabilities. OH CH
-
Read the following and then answer the questions below:September 12: A Brisbane business offers by letter to sell 500 tyres to a New Zealand company. The Brisbane company does not specify a method of...
-
Fred returns home from work one day to discover his house surrounded by police. His wife is being held hostage and threatened by her captor. Fred pleads with the police to rescue her and offers...
-
Would like you to revisit one of these. Consideration must be clear and measurable.if you can't measure it then how can you show it has / has not been done?How can you sue someone for breach of...

Study smarter with the SolutionInn App


