Identify the Michael donor and Michael acceptor that could be used to prepare each of the following
Question:
(a)
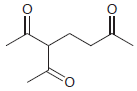
(b)
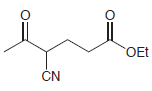
(c)
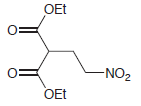
(d)
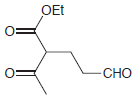
(e)
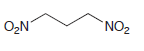
Transcribed Image Text:
OEt ČN
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (10 reviews)
a b c d e ...View the full answer

Answered By

JAPHETH KOGEI
Hi there. I'm here to assist you to score the highest marks on your assignments and homework. My areas of specialisation are:
Auditing, Financial Accounting, Macroeconomics, Monetary-economics, Business-administration, Advanced-accounting, Corporate Finance, Professional-accounting-ethics, Corporate governance, Financial-risk-analysis, Financial-budgeting, Corporate-social-responsibility, Statistics, Business management, logic, Critical thinking,
So, I look forward to helping you solve your academic problem.
I enjoy teaching and tutoring university and high school students. During my free time, I also read books on motivation, leadership, comedy, emotional intelligence, critical thinking, nature, human nature, innovation, persuasion, performance, negotiations, goals, power, time management, wealth, debates, sales, and finance. Additionally, I am a panellist on an FM radio program on Sunday mornings where we discuss current affairs.
I travel three times a year either to the USA, Europe and around Africa.
As a university student in the USA, I enjoyed interacting with people from different cultures and ethnic groups. Together with friends, we travelled widely in the USA and in Europe (UK, France, Denmark, Germany, Turkey, etc).
So, I look forward to tutoring you. I believe that it will be exciting to meet them.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Which diene and which dienophile could be used to prepare each of the following compounds? a. b. c. d. 1 CH3 Cl CI
-
Show how a conjugate addition can be used to prepare each of the following compounds. (a) 3,4-dimethyl-2-hexanone (2 ways) (b) levulinic acid
-
Show how a Wittig reaction can be used to prepare each of the following compounds. In each case, also show how the Wittig reagent would be prepared: (a) (b)
-
An orange grower in Florida faces a dilemma. The weather forecast is for cold weather, and there is a 50% chance that the temperature tonight will be cold enough to freeze and destroy his entire...
-
For the plane stress element shown in Figure P15 -14 subjected to a uniform temperature rise of T = 50°C, determine the thermal force matrix {fT}. Let E = 70 GPa, v = 0.3, α = 23...
-
creating a email about a Easter bunny costume with a preheader and call to action and subject
-
An increase in the capital gains tax rate _________ unrealized returns.
-
Explain how a sales order, a production order, a materials requisition form, and a labor time ticket are involved in producing and costing products?
-
Please fill in the blank. 1. An insurance company which is owned by its policyholders is called a _____________ 2. The lag between the time that coverage is sold and the time when claims are paid is...
-
Jimmy owns a garden in which he has planted N trees in a row. After a few years, the trees have grown up and now they have different heights. Jimmy pays much attention to the aesthetics of his...
-
A(n) ______ ______ design is one in which each experimental unit is randomly assigned to a treatment. A(n) ______ ______ design is one in which the experimental units are paired up.
-
1. True or False: Observational studies are used to determine causality between explanatory and response variables. 2. True or False: Generally, the goal of an experiment is to determine the effect...
-
Imagine a hypothetical country of 100 million ruled by a ruthless dictator bent on suppressing opposition. The opposition is estimated at 1000 individuals, all living in exile. The dictatorship is...
-
Dr. Kovaleski is interested in examining whether quantity of sleep impacts problem solving ability. To test problem solving ability, the research team gave participants a puzzle and measured how long...
-
Can you please help me fill out the spreadsheet? Idexo Corporation is a privately held designer and manufacturer of licensed college apparel in Cincinnati, Ohio. In late 2020, after several years of...
-
CHECK FIGURE: Adjusted book balance = $2,837.06 Mae Telford, the controller of the Baylor Company, provided the following information: Bank statement balance Add: Baylor Company Bank Reconciliation...
-
Read the Scenario Congratulations, you are now the Police Chief in Anytown, USA. A city with 30,000 residents and you are responsible to provide 24 hour a day police coverage. You have a total of 45...
-
Here are summary statistics for randomly selected weights of newborn girls: n = 36, x = 3180.6 g, s = 700.5 g. Use a confidence level of 99% to complete parts (a) through (d) below. a. Identify the...
-
Two cars leave from the same point at the same time, traveling in opposite directions. One travels 15 mph slower than the other. After 6 hr, they are 630 mi apart. Find the rate of each car. Starting...
-
Factor and simplify, if possible. Check your result using a graphing calculator. 3 cot 2 + 6 cot + 3
-
Give the product formed when the following alcohols is oxidized by dilute HNO3. HOCH2CH 2CH2OH
-
Assuming you knew the properties of the compound obtained in Problem 24.23, including its optical rotation, show how you could use periodate oxidation to distinguish methyl -D- galactopyranoside from...
-
Assuming you knew the properties of the compound obtained in Problem 24.23, including its optical rotation, show how you could use periodate oxidation to distinguish methyl -D- galactopyranoside from...
-
September 23 for $1,050 each. On December 24 , it sold one of the diamonds that was purchased on July 9 . Using the specific identification method, its ending inventory (after the December 24 sale)...
-
Madsen Motors's bonds have 13 years remaining to maturity. Interest is paid annually, they have a $1,000 par value, the coupon interest rate is 8%, and the yield to maturity is 10%. What is the...
-
Builder Products, Incorporated, uses the weighted - average method in its process costing system. It manufactures a caulking compound that goes through three processing stages prior to completion....

Study smarter with the SolutionInn App


