Show how a Wittig reaction can be used to prepare each of the following compounds. In each
Question:
(a)
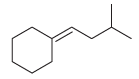
(b)
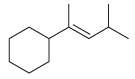
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 84% (13 reviews)
a b ...View the full answer

Answered By

Fahmin Arakkal
Tutoring and Contributing expert question and answers to teachers and students.
Primarily oversees the Heat and Mass Transfer contents presented on websites and blogs.
Responsible for Creating, Editing, Updating all contents related Chemical Engineering in
latex language
4.40+
8+ Reviews
22+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Indicole which of the method in this section could be used to prepare each of the following amino acids. For each method that can be used, give an equation. For each case in which a method would not...
-
Which diene and which dienophile could be used to prepare each of the following compounds? a. b. c. d. 1 CH3 Cl CI
-
Identify the Michael donor and Michael acceptor that could be used to prepare each of the following compounds via a Michael addition. (a) (b) (c) (d) (e) OEt N
-
Bloomberg Intelligence listed 50 companies to watch in 2018 (www .bloomberg.com/features/companies-to-watch-2018). Twelve of the companies are listed here with their total assets and 12-month sales....
-
On November 1 2014, Fain Corporation paid principal and interest on a 6 month, 8% note payable with a face amount of $5000. How did this affect Fain financial statement? Exp Liabilities Stk Rev Net...
-
A population grows according to the logistic law with a limiting population of 5 x 109 individuals. The initial population of 0.2 x 109 begins growing by doubling every hour. What will the population...
-
What is the character of a bond issuer?
-
The Good'n'Fresh Grocery Store has two checkout lanes and four employees. Employees are equally skilled, and all are able to either operate a register (checkers) or bag groceries (baggers). The store...
-
Net Present ValueUnequal Lives Project 1 requires an original investment of $70,800. The project will yield cash flows of $14,000 per year for eight years. Project 2 has a calculated net present...
-
Mazud is a BC resident who has been involved with a part-time entrepreneurship activity for the past two years. He maintains good relations with certain local restaurants and his customers to sustain...
-
Draw the structure of the alkyl halide needed to prepare each of the following Wittig reagents, and then determine which Wittig reagent will be the more difficult to prepare. Explain your choice: Ph...
-
Choose a Grignard reagent and a ketone that can be used to produce each of the following compounds: (a) 3-methyl-3-pentanol (b) 1-ethylcyclohexanol (c) Triphenylmethanol (d) 5-phenyl-5-nonanol
-
This information is available for PepsiCo, Inc. for 2004, 2005, and 2006. Instructions Calculate the inventory turnover ratio , days in inventory, and gross profit rate for PepsiCo., Inc. for 2004,...
-
Wedona Energy Consultants prepares adjusting entries monthly. Based on an analysis of the unadjusted trial balance at January 31, 2014, the following information was available for the preparation of...
-
K Archeologists have studied sizes of Egyptian skulls in an attempt to determine whether breeding occurred between different cultures. Listed below are the widths (mm) of skulls from 150 A.D....
-
The Farm Financial Standards Council (FFSC) Multiple Choice recommends uniform procedures and standards for preparing farm financial statements. reviews loan applications. sets financial goals for...
-
An alphabetical list of the adjusted trial balance accounts for North Country Rentals after its first year of operations ending March 31, 2023, is shown below. Accounts payable Account Accumulated...
-
Renting SUVs The airport branch of a car rental company maintains a fleet of 50 SUVs. The airport branch is open 24 hours a day. Customers arrive at the rental company from the airport 24 hours a day...
-
The College of Central North (a small private not-for-profit entity) has the following events and transactions. a. On January 1, Year 1, the board of trustees votes to restrict $1.9 million of...
-
A researcher reports a significant two-way between-subjects ANOVA, F(3, 40) = 2.96. State the decision to retain or reject the null hypothesis for this test.
-
Classify each of the following objects as to whether it is chiral or achiral: (a) A screwdriver (b) A baseball bat (c) A golf club (d) A tennis shoe (e) An ear (f) A woodscrew (g) A car (h) A hammer
-
Give (R) and (S) designations for each pair of enantiomers given as answers to Practice Problem 5.4. In problem 5.4 (c) 2-Chlorobutane (d) 2-Methyl-1-butanol (f) 2-Bromopentane (h) 3-Methylhexane (j)...
-
List the substituents in each of the following sets in order of priority, from highest to lowest: (a) -Cl, -OH, -SH, -H (b) -CH3, -CH2Br, -CH2Cl, -CH2OH (c) -H, -OH, -CHO, -CH3 (d) -CH(CH3)2,...
-
A partnership agreement provides that half of the annual profit be distributed in proportion to each partners investment in the partnership, and that the other half be distributed in proportion to...
-
Employee deduction calculation: 6.45% for 2021 Question 2 options: True False
-
Paula Boothe, president of the Sarasota Corporation, has mandated a minimum 6% return on investment for any project undertaken by the company. Given the companys decentralization, Paula leaves all...
Cycles Of Spin Strategic Communication In The US Congress 1st Edition - ISBN: 052113580X - Free Book

Study smarter with the SolutionInn App


