Identify the reagents you would use to accomplish each of the following transformation. a. b. c. d.
Question:
a.
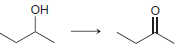
b.
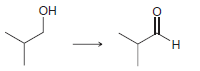
c.
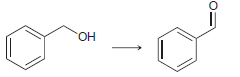
d.
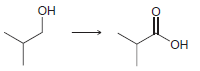
e.
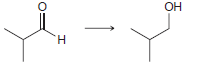
f.
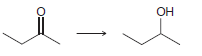
Transcribed Image Text:
Он ОН H.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 68% (16 reviews)
a b c ...View the full answer

Answered By

Allan Simiyu
I am an adroit Writer. I am a dedicated writer having worked as a writer for 3 years now. With this, I am sure to ace in the field by helping students break down abstract concepts into simpler ideas.
5.00+
8+ Reviews
54+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Identify the reagents you would use to convert cyclohexanone into each of the following compounds. (a) (b) (c) (d) (e) (f) (g)
-
Identify the reagents you would use to accomplish the following transformation - Br Br
-
Identify the reagents you would use to accomplish each of the following transformations: a. b.
-
In a new margin account, an investor sells short 1,000 shares of XYZ stock at $80 per share. If the market value of the stock falls to $70, if the leverage ratio is 1.53846 1. what is the initial...
-
Refer to Scenario 3. The accuracy of a labor standard is to be within 5%, and the confidence level is 95% (Z = 1.96). What is the approximate sample size for Element 1? Scenario 3 The data in the...
-
Find both first partial derivatives. (x, y) = 2x + y
-
Choose the final price(s).
-
Miller Corporation is considering replacing a machine. The replacement will reduce operating expenses (that is, increase earnings before depreciation, interest, and taxes) by $16,000 per year for...
-
Khanna Inc. had the following transactions, information and balances in its accounting records for fiscal 2021: (CAD = Canadian dollars; USD = U.S. dollars) Balance (CAD) Dec 31/21 CAD treasury bill...
-
Allie has bought a new apple orchard. The orchard has a single file of trees, numbered from 1 to N. Each tree has a certail number of ripe apples. Allie has a rule she wants to follow. She wants to...
-
Draw the mechanism for each of the following reaction. a. b. c. OH py
-
Predict the products for each of the following: a. b. c. d. e. f. 1) O3 2) DMS 3) Excess LAH 4) H20 1) O3 2) DMS 3) Excess LAH 4) H20
-
Compare the pros and cons of using a network structure to perform organizational activities versus performing all activities in-house or within one organizational hierarchy. [LO18-3, 18-4, 18-5]
-
Systems thinking is all about solving problemsin organizations, world situations, and even our personal lives. But it is not just a procedure; it is a different way of approaching problems. Our...
-
How would I display the following 3 principles in an entertaining infographic? Be very specific . Principle 1: Employee Engagement and Motivation Drawing from the Human Relations Movement theory and...
-
Shown below is a cross section of tubular member which is subjected to a torque T= 5.5 kN-m. It has a length L-3.0-m and the material shear modulus G=27 GPa. Dimensions: b=150 mm, h= 100 mm and t= 8...
-
The hip roof shown in the below Figure 2 is constructed of 2x10 rafters spaced 16 inches on center. The hip rafters are 1 -inch-wide by 12-inch-high GLBs. The roof has a slope of 4:12. Prepare a list...
-
2. Estimate the populations of Fargo, ND and Bismarck, ND in years of 2040 and 2050. Select a single value of population that you would use for design purposes in each year. You need to specify and...
-
The amount of heat transferred per hour through a glass window varies jointly with the surface area of the window and the difference in temperature between the areas separated by the glass. A window...
-
Why should you not model a decision variable as a random variable with a probability distribution?
-
Draw structures of glyceryl tripalmitate and glyceryl trioleate. Which would you expect to have a higher melting point?
-
Draw the structure of magnesium oleate, a component of bathtub scum.
-
Write the saponification reaction of glyceryl di-oleate monopalmitate with aqueous NaOH.
-
Domino is 4 0 years old and is married out of community of property with the exclusion of the accrual system to Dolly ( 3 5 ) . They have one child, Domonique, who is 1 1 years old. Domino resigned...
-
YOU ARE CREATING AN INVESTMENT POLICY STATEMENT FOR JANE DOE General: 60 years old, 3 grown children that are living on their own and supporting themselves. She is in a very low tax rate so we don't...
-
firm purchased a new piece of equipment with an estimated useful life of eight years. The cost of the equipment was $65,000. The salvage value was estimated to be $10,000 at the end of year 8. Using...

Study smarter with the SolutionInn App


