Identify the reagents you would use to achieve each of the following transformations: (a) (b)
Question:
Identify the reagents you would use to achieve each of the following transformations:
(a)
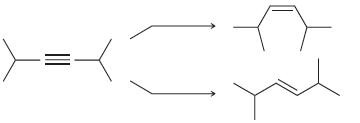
(b)
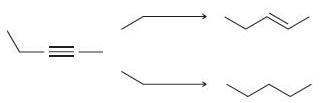
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 63% (11 reviews)
a b H ...View the full answer

Answered By

JAPHETH KOGEI
Hi there. I'm here to assist you to score the highest marks on your assignments and homework. My areas of specialisation are:
Auditing, Financial Accounting, Macroeconomics, Monetary-economics, Business-administration, Advanced-accounting, Corporate Finance, Professional-accounting-ethics, Corporate governance, Financial-risk-analysis, Financial-budgeting, Corporate-social-responsibility, Statistics, Business management, logic, Critical thinking,
So, I look forward to helping you solve your academic problem.
I enjoy teaching and tutoring university and high school students. During my free time, I also read books on motivation, leadership, comedy, emotional intelligence, critical thinking, nature, human nature, innovation, persuasion, performance, negotiations, goals, power, time management, wealth, debates, sales, and finance. Additionally, I am a panellist on an FM radio program on Sunday mornings where we discuss current affairs.
I travel three times a year either to the USA, Europe and around Africa.
As a university student in the USA, I enjoyed interacting with people from different cultures and ethnic groups. Together with friends, we travelled widely in the USA and in Europe (UK, France, Denmark, Germany, Turkey, etc).
So, I look forward to tutoring you. I believe that it will be exciting to meet them.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Identify the reagents you would use to convert cyclohexanone into each of the following compounds. (a) (b) (c) (d) (e) (f) (g)
-
Identify what reagents you would use to achieve each transformation: a. Conversion of 2-methyl-2-butene into a secondary alkyl halide b. Conversion of 2-methyl-2-butene into a tertiary alkyl halide...
-
Identify the reagents you would use to convert 2-bromo-2-methylbutane into 3-methyl-1-butyne.
-
answer the following questions about the countries below using CIA factbook Economic Systems Continuum Activity: Country Information Cards Answer the following questions about the countries below...
-
What is the main argument in the article, Global Metropolis: The Role of Cities and Metropolitan Areas in the Global Economy.
-
Find to four significant digits for 0 < 2. csc = 3.940
-
Do you have a training program?
-
Desrosiers Ltd. had the following long-term receivable account balances at December 31, 2013: Notes receivable........ $1,800,000 Notes receivable-Employees... 400,000 Transactions during 2014 and...
-
[ The following information applies to the questions displayed below. ] Arboles Company manufactures pencils and has the following information available for the month of July: Work in process, July 1...
-
V.T. a 34-year-old woman, presents complaining of fatigue. difficulty concentrating, and a decline in memory She also notes unexplained weight gain over the past six months, an abnormal loss of hair,...
-
Describe the Six Sigma processes and how they came about. How is Six Sigma related to lean thinking?
-
Use the relation the cyclic rule and the van der Waals equation of state, to derive an equation for C P,m C V,m in terms of V m , T, and the gas constants R, a, and b. aV . Cr m = T
-
Data for Nosker Company are presented in P17.7A. Further analysis reveals the following. 1. Accounts payable pertain to merchandise suppliers. 2. All operating expenses except for depreciation were...
-
List and describe three common interview mistakes which compromise the quality of the selection process.
-
Player I chooses a positive integer x > 0 and player II chooses a positive integer y > 0. The player with the lower number pays a dollar to the player with the higher number unless the higher number...
-
1. Distinguish between risk management and risk transfer. As the manager of XYZ FBO, what measures would you put in place to ensure risk reduction?
-
Devlop a sumary of your understanding of Learning and Retention. In the sumary, think about how you see Learning and Retention fitting into the entire study of Organizational Behavior. How does the...
-
Emani, Peters and Desai et al (2018) "conducted a cross-sectional survey of adopters and non-adopters of the portal...the survey consisted of perceived attributes from the DOI theory,...
-
Perform the indicated operations. m3m + 5m - 3 m - 1
-
l ask this second time correnct answer is 38,01 can we look pls Consider a non-conducting rod of length 8.8 m having a uniform charge density 4.5 nC/m. Find the electric potential at P, a...
-
A Which reaction in each of the following pairs would you expect to be faster? (a) The S N 2 displacement by I on CH 3 CI or on CH 3 OTos (b) The SN2 displacement by CH 3 CO 2 on bromomethane or on...
-
What products would you expect from the reaction of 1-bromopropane with each of the following? (a) NaNH2 (b) KOC (CH3)3 (c) NaI (d) NaCN (e) NaC CH (f) Mg, then H2O
-
Which reactant in each of the following pairs is more nucleophilic? Explain. (a) NH2 or NH3 (b) H2O or CH3CO2 (c) BF3 or F (d) (CH3)3P or (CH3)3N (e) I or Cl (f) C N or OCH3
-
Discuss American History
-
Your firm has developed a new lithium ion battery polymer that could enhance the performance of lithion ion batteries. These batteries have applications in many markets including cellphones, laptops,...
-
Need help analyzing statistical data 1. ANOVA) True or false: If we assume a 95% confidence level, there is a significant difference in performance generally across all groups. 2. (t-test) True or...

Study smarter with the SolutionInn App


