In each of the following transformations, identify whether the starting material has been oxidized, reduced, or neither.
Question:
a.
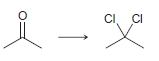
b.
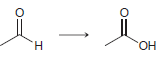
c.
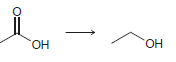
d.
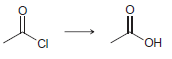
e.
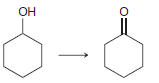
f.
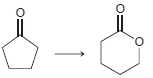
CI CI но. ОН H.
Step by Step Answer:
a 2 2 the starting material is neither oxidized nor redu...View the full answer



Related Video
Lemon juice preserves apples by slowing down the oxidation process. Oxidation is a chemical reaction that occurs when oxygen reacts with certain substances, such as apples. When an apple is cut or bitten, oxygen is exposed to the inside of the apple and causes enzymes in the apple to turn brown, which is an indication of oxidation. The browning process is caused by the production of polyphenol oxidase (PPO) enzymes that convert phenolic compounds into quinones, which then polymerize to form the brown pigments. One of the compounds present in lemon juice is ascorbic acid (vitamin C), which is a natural antioxidant. Antioxidants work by neutralizing the free radicals that cause oxidation. When lemon juice is applied to apples, the ascorbic acid in the lemon juice reacts with the PPO enzymes and slows down the browning process. You can do an experiment by cutting apples into small pieces, leaving one apple piece in contact with air and the others covered with lemon juice and compare the browning process. This will help to understand the antioxidation process in fruits.
Students also viewed these Sciences questions
-
Determine whether refrigerant R-22 in each of the following states is a compressed liquid, a superheated vapor, or a mixture of saturated liquid and vapor.
-
Calculating Break-Even in each of the following cases, calculate the accounting break-even and the cash break-even points. Ignore any tax effects in calculating the cash break-even. Unit Price Unit...
-
Determine whether the taxpayer in each of the following situations has a claim of right to the income received: a. Sulley's Spa Spot sells hot tubs that have a 2-year warranty. The warranty provides...
-
List at least one "real life" situation that may arise out of each category. Be sure to explain how you think each issue should be handled, policy-wise. 1. Relationships with citizens and others over...
-
A person starts work for a retail company and is treated reasonably well, at first. But after a few years she notices the company is suddenly putting a lot more demands on her and in other ways is...
-
Student researchers wanted to see whether listening to music with lyrics would affect a persons ability to memorize words compared to listening to music without lyrics. They recruited 31 people for...
-
decide if the events shown in the Venn diagram are mutually exclusive. Explain your reasoning.
-
Researchers developed a questionnaire to identify compulsive buyers. A random sample of 32 subjects who identified themselves as compulsive buyers was obtained, and they had a mean questionnaire...
-
Use the following to answer questions (2) through (5): Suppose the number of cases of COVID-19 (labeled CV) in a particular geographic region depends on the number of face masks purchased in the...
-
Cordova Research Group spent more than thirty years conducting pure and applied research for a variety of external customers. With the reduction, however, in R&D funding, Cordova decided that the...
-
There are only four constitutional isomers with molecular formula C 4 H 9 NO 2 that contain a nitro group (-NO 2 ). Three of these isomers have similar pK a values, while the fourth isomer has a much...
-
Cyclopropane is a compound in which the carbon atoms form a three-membered ring: Each of the carbon atoms in cyclopropane is sp 3 hybridized. Cyclopropane is more reactive than other cyclic compounds...
-
The following were selected from among the transactions completed by Southmont Company during April of the current year: {Instructions} Journalize the transactions.obj. 3 Apr. 3. Purchased...
-
What is judgement sampling?Explain with a suitable example.
-
What is convenience sampling?Explain with a suitable example.
-
Customer Distribution Channels (all amounts in thousands of U.S. Dollars) Wholesale Customers Retail Customers Total Total N. America S. America Total Wholesale Wholesaler Wholesaler Retail Green...
-
MA Assignment 3 Motor Tyres manufactures one size of tyre in each of its production lines. The following information relates to one production line for the most recent period. The company uses the...
-
Describe an operational unit within an organization, and define the mission statement, goals, and operational objectives for the unit. The operational unit should be an organization they would like...
-
The following table lists projected global virtual reality head-mounted display shipments H in millions of units x years after 2015. (a) Find a function H(x) that models the data. (b) Graph H and the...
-
Some people argue that the internal control requirements of the Sarbanes-Oxley Act (SOX) put U.S. companies at a competitive disadvantage to companies outside the United States. Discuss the...
-
Show the products obtained from addition of methyl magnesium bromide to the following compounds: (a) Cyclopentanone (b) Benzophenone (diphenyl ketone) (c) 3-Hexanone
-
Use a Grignard reaction to prepare the following alcohols: (a) 2-Methyl-2-propanol (b) 1-Methylcyclohexanol (c) 3-Methyl-3-pentanol (d) 2-Phenyl-2-butanol (e) Benzyl alcohol (f) 4-Methyl-1-pentanol
-
Use the reaction of a Grignard reagent with a carbonyl compound to synthesize the following compound:
-
When preparing government-wide financial statements, the modified accrual based governments funds are adjusted. Please show the adjustments (in journal entry form with debits and credits) that would...
-
I need help finding the callable price and call value
-
On 31 October 2022, the owner took goods for his son as a birthday gift. The cost price of the goods was R15 000

Study smarter with the SolutionInn App


