In each reaction, identify the Lewis acid and the Lewis base: (a) (b) (c) F L-
Question:
(a)
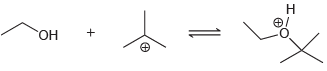
(b)
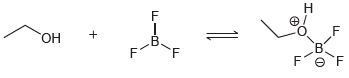
(c)
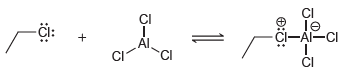
Transcribed Image Text:
Н ОН Н ОН он ӨF L-Ф
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 73% (15 reviews)
I n each reaction b...View the full answer

Answered By

Mustafa olang
Please accept my enthusiastic application to solutionInn. I would love the opportunity to be a hardworking, passionate member of your tutoring program. As soon as I read the description of the program, I knew I was a well-qualified candidate for the position.
I have extensive tutoring experience in a variety of fields. I have tutored in English as well as Calculus. I have helped students learn to analyze literature, write essays, understand historical events, and graph parabolas. Your program requires that tutors be able to assist students in multiple subjects, and my experience would allow me to do just that.
You also state in your job posting that you require tutors that can work with students of all ages. As a summer camp counselor, I have experience working with preschool and kindergarten-age students. I have also tutored middle school students in reading, as well as college and high school students. Through these tutoring and counseling positions, I have learned how to best teach each age group. For example, I created songs to teach my three-year-old campers the camp rules, but I gave my college student daily quizzes to help her prepare for exams.
I am passionate about helping students improve in all academic subjects. I still remember my excitement when my calculus student received her first “A” on a quiz! I am confident that my passion and experience are the qualities you are looking for at solutionInn. Thank you so much for your time and consideration.
4.80+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
(a) Identify the Brønsted-Lowry acid and base in the reaction (b) Identify the Lewis acid and base in the reaction. = H = N
-
(a) The following diagram represents the reaction of PCl4+ with Cl-. Draw the lewis structures for the reactants and products, and identify the lewis acid and the lweis base in the reaction. (b) The...
-
In each case below, identify the Lewis acid and the Lewis base: (a) (b)
-
Match each of the following energy band structures with the type of material it represents. Show Work A B C Empty conduction band Band gap Filled valence band Empty conduction band Band gap Filled...
-
Should Mexican trucks be permitted to haul goods freely into the United States? In presenting your views be sure to include NAFTA, transportation safety, illegal drugs, and illegal immigration.
-
Determine the horizontal force F required to maintain equilibrium of the slider mechanism when = 60. Set M = 6 N m D 0.5 m A F 0.5 m B M S 0.5 m
-
It is possible for an organization to add value through overseas operations? L01
-
The following amounts were taken from the weekly payroll register for the Wu Vale Company on August 9, 201X. Please prepare the general journal entry to record the payroll for the Wu Vale Company for...
-
Rouse Recliners manufactures leather recliners and uses flexible budgeting and a standard cost system. Rouse Recliners allocates overhead based on yards of direct materials. The company's performance...
-
Two metals A (melting point 800C) and B (melting point 650C) form a binary isomorphous system. An alloy containing 45 wt% metal B is found to consist of 75 wt% solid and the remainder liquid whereas...
-
Determine the relationship between the two structures below. Are they resonance structures or are they constitutional isomers?
-
Trans-1,3-Dichlorocyclobutane has a measurable dipole moment. Explain why the individual dipole moments of the C-Cl bonds do not cancel each other to produce a zero net dipole moment. CI-...
-
Elite Apparel purchased 80 new shirts for cash and recorded a total cost of \(\$ 2,620\), determined as follows: Required: Make the needed corrections in this calculation. Prepare the journal...
-
Factor the expression. 4x+31x+21
-
What was the total cost of Job #1253 for January? * (1 Point) BREAD Co. is a print shop that produces jobs to customer specifications. During January 2019, Job #1253 was worked on and the following...
-
The Greensboro Performing Arts Center (GPAC) has a total capacity of 7,600 seats: 2,000 center seats, 2,500 side seats, and 3,100 balcony seats. The budgeted and actual tickets sold for a Broadway...
-
eBook Current position analysis The bond indenture for the 10-year, 9% debenture bonds issued January 2, 2015, required working capital of $100,000, a current ratio of 1.5, and a quick ratio of 1 the...
-
Explain Below terms 1-Leverage Ratios 2-Profitability Ratios 3-Market Value Ratios 4-Liquidity Ratios 5-Efficiency Ratios
-
Write a system of linear equations with two variables whose solution satisfies the problem. State what each variable represents. Then solve the system. Admission prices to a movie are $4 for children...
-
Create a data model for one of the processes in the end-of-chapter Exercises for Chapter 4. Explain how you would balance the data model and process model.
-
Build a handheld model of bicycle [2.2.1] heptanes and discuss the types of strain that are present in this compound. udy tee op!
-
Explain whether the compound shown is the Z or the E diastereomer. Problems using online Three-Dimensional molecular models
-
Explain whether the conformation shown is the most stable conformation of each of these molecules. Problems using online Three-Dimensional molecular models
-
You have just been hired as a new management trainee by Earrings Unlimited, a distributor of earrings to various retail outlets located in shopping malls across the country. In the past, the company...
-
Brief Exercise 10-6 Flint Inc. purchased land, building, and equipment from Laguna Corporation for a cash payment of $327,600. The estimated fair values of the assets are land $62,400, building...
-
"faithful respresentation" is the overriding principle that should be followed in ones prepaparation of IFRS-based financial statement. what is it? explain it fully quoting IAS. how this this...

Study smarter with the SolutionInn App


