Predict the major product(s) for each of the following thermal electrocyclic reactions: (a) (b) (c) Heat Heat
Question:
(a)
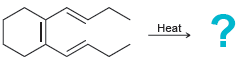
(b)
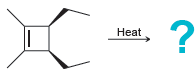
(c)
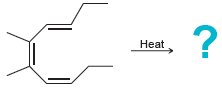
Transcribed Image Text:
Heat Heat
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (12 reviews)
a b...View the full answer

Answered By

Gilbert Chesire
I am a diligent writer who understands the writing conventions used in the industry and with the expertise to produce high quality papers at all times. I love to write plagiarism free work with which the grammar flows perfectly. I write both academics and articles with a lot of enthusiasm. I am always determined to put the interests of my customers before mine so as to build a cohesive environment where we can benefit from each other. I value all my clients and I pay them back by delivering the quality of work they yearn to get.
4.80+
14+ Reviews
49+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Predict the major product (or products) formed when each of the following reacts with a mixture of concentrated HNO3 and H2SO4. (a) (b) (c) 4-Chlorobenzoic acid (d) 3-Chlorobenzoic acid (e)...
-
Predict the major product from each of the following reactions. (a) (b) (c) (d) (e) (f) (g) (h) (i) CH NH2, cat. HA N-H, cat. HA NH2 cat. HA PPha (1) HS SH (2) Raney Ni, H2 0 CH2PPha (excess) O. O...
-
Predict the major product from each of the following reactions. (a) (b) (c) (d) (e) HO OH NH2 HA (cat.) (1) HCN (2) LiAIH4 (3) H2o mCPBA
-
If the interest rate on debt is lower than ROA, then a firm will Valitse yksi: a. increase ROE b. not change ROE c. none of these is correct d. decrease ROE e. change the ROE in an indeterminable...
-
The following data were obtained from a CU test with pore pressures measured on an undisturbed specimen of sandy silt. The consolidation pressure was 850 kPa and the specimen was sheared in axial...
-
A thick piece of conducting wire is bent into a circle of radius a. At the center of the circle (point P) is a pivot on which rests a conducting bar of length a. The bar makes electrical contact with...
-
Listed here are three different products in various stages of the product life cycle. What marketing strategies would you suggest to these companies? (a) Canon digital camerasgrowth stage, (b)...
-
Compute cost of goods sold for year 2013 using the following information. Finished goods inventory, Dec. 31, 2012 . . . . . . . . . . . . $ 345,000 Goods in process inventory, Dec. 31, 2012 . . . . ....
-
Question 1 points Classify the following activities as operating, investing, financing or noncash financing and investing activibes Select an activity by clicking to the nght of each cash flow,...
-
A workday begins each morning at 8 A. M. in the tax assessors office. The staff is composed of the director, two secretaries, two computer- typists, and three file clerks. Until last year, the office...
-
Consider the following [4+4] cycloaddition process. Would you expect this process to occur through a thermal or photochemical pathway? Justify your answer with MO theory. +
-
Assume the following values for Figures 4.4a and 4.4b. Q 1 = 20 bags. Q 2 = 15 bags. Q 3 = 27 bags. The market equilibrium price is $45 per bag. The price at a is $85 per bag. The price at c is $5...
-
In Exercises find a polar equation for the conic with its focus at the pole. (For convenience, the equation for the directrix is given in rectangular form.) Conic Parabola Vertex or Vertices ) 2 1,
-
Silver Company makes a product that is very popular as a Mothers Day gift. Thus, peak sales occur in May of each year, as shown in the companys sales budget for the second quarter given below: April...
-
Among the following statements, select the ones which have a positive environmental impact. Choose several answers Minimising the impact of a product on the environment Avoiding the destruction of a...
-
Developing Financial Statements: All organizations, including those in the healthcare industry, need to make money to be profitable and survive. Financial statements, such as balance sheets, profit...
-
The engineers estimated that on average, fuel costs, assuming existing routes and number of flights stay the same, would decrease by almost 18% from an average of 42,000 gallons of jet fuel per...
-
It's the latest Berkeley trend: raising chickens in a backyard co-op coop. (The chickens cluck with delight at that joke.) It turns out that Berkeley chickens have an unusual property: their weight...
-
In Problems 914, find the domain of each function. f(x) = 2 - x
-
Why do markets typically lead to an efficient outcome for buyers and sellers?
-
Treatment of 4-penten-l-ol with aqueous Br2 yields a cyclic bromo ether rather than the expected bromohydrin. Suggest a mechanism, using curved arrows to show electronmovement. CH2B Br2, H20 %3...
-
Hydroboration of 2-mnethyl-2-pentene at 25?C followed by oxidation with alkaline 11202 yields 2-methyl-3-pentanol, but hydroboration at 160?C followed by oxidation yields 4-methyl?1?pentanol. Suggest...
-
We?ll see in the next chapter that alkynes undergo many of the same reactions that alkenes do. What product might you expect from each of the following reactions? 1 equiv Br2 CH3 (a) 2 equiv H2, Pd/C...
-
Docs Auto Body has budgeted the costs of the following repair time and parts activities for 2009: Doc's budgets 6,000 hours of repair time in 2009. A profit margin of $7 per labour hour will be added...
-
QUESTION 28 In a perpetual inventory system, the cost of inventory sold is: Debited to accounts receivable. Debited to cost of goods sold. O Not recorded at the time goods are sold. O Credited to...
-
The following financial statements and additional information are reported. IKIBAN INC. Comparative Balance Sheets June 30, 2019 and 2018 2019 2018 $105,709 69,500 66,800 4,700 246,700 127,eee...

Study smarter with the SolutionInn App


