Predict the stereochemical outcome for each of the following E2 reactions. In each case, draw only the
Question:
a.
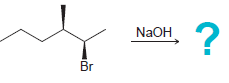
b.
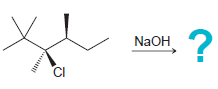
Transcribed Image Text:
? NaOH Br NaOH CI
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (10 reviews)
a b...View the full answer

Answered By

OTIENO OBADO
I have a vast experience in teaching, mentoring and tutoring. I handle student concerns diligently and my academic background is undeniably aesthetic
4.30+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Use your answers to the preceding problem to predict the stereochemical outcome of the addition of bromine to maleic acid and to fumaric acid. (a) Which dicarboxylic acid would add bromine to yield a...
-
Predict the stereochemical outcome of radical bromination of the following alkanes: (a) (b) (c) (d)
-
Predict the major product for each of the following E2 reactions: a. b. c. d. NaOH Br NaOH Br
-
What is the "ALDI Way" and what was its quest? Take out costs; eliminate complexity Survival; make enough money to pay overhead costs Increase market share; find more prospective customers Build...
-
A Christian Philosophical journey by Schenck kenneth In a Word document, respond to all the following questions: How do I know what is true? Can I trust my senses? Can I trust my mind? Does God...
-
Find the slope and y-intercept of the line with the given equation and sketch the graph using the slope and y-intercept. A calculator can be used to check your graph. y = 4x
-
Comparing hand strength. Is the right hand generally stronger than the left in right-handed people? You can crudely measure hand strength by placing a bathroom scale on a shelf with the end...
-
Waymon Co. has net sales of $100,000, cost of goods sold of $70,000, and operating expenses of $18,000. What is its gross profit?
-
please do all parts to the question! 3. AT&T provides long-distance telephone service in Pleasantville. According to the company's records, the average length of all long-distance calls placed...
-
11.9. Town and Country has experienceda substantial increase in business volume because of recent fare wars between the major air carriers. Town and Country operates a single office at a major...
-
Some energy experts refer to the Great Plains states as the Saudi Arabia of wind power. Explain what the reference means.
-
Japan wishes to make use of solar power, but it does not have extensive tracts of land for building large solar power plants. Which solar technology do you think is best suited to Japan's needs? Why?
-
With the other members of your group, create a list of software frequently used (at school and work) by each member of the team. Identify which sphere of influence each software supports. Create a...
-
Solve for "C" and "E": 1) E cos (15)-C=0 2) -300+ C+E sin (15) = 0
-
Let u=3, b. Compute uv, uv, 2-3 v =
-
Using Complex Numbers show that d cosz=-sinz dz
-
use for loops to solve the following problems 1. Write a complete C++ program that does the following. It asks the user to enter their age (which is assumed to be a positive integer). The program...
-
Profile Vickers hardness test Penetrating body: Square diamond pyramid :Test force F N ... 981 N (HV 5 ... HV 100) 49 :Measured value Diagonals of the square impression d Hardness value: F 0,189 F...
-
Simplify each expression. Assume that all variables represent nonzero real numbers. (y-x4)-3
-
The 2017 financial statements of the U.S. government are available at: https://www.fiscal.treasury.gov/fsreports/rpt/finrep/fr/fr_index.htm Use these to answer the following questions: a. Statement...
-
The following reaction involves a conjugate addition reaction followed by an intra molecular Claisen condensation. Write both steps, and show theirmechanisms. CO2CH3 (CH3i>CuLi -CEC-CO2CH3 H
-
The following reaction involves two successive intra molecular Michael reactions. Write both steps, and show theirmechanisms. Nat "OEt Ethanol
-
The following reaction involves an intra molecular aldol reaction followed by a ret ro aldol-like reaction. Write both steps, and show theirmechanisms. O2Et CO2Et Na* "OEt Ethanol
-
When preparing government-wide financial statements, the modified accrual based governments funds are adjusted. Please show the adjustments (in journal entry form with debits and credits) that would...
-
I need help finding the callable price and call value
-
On 31 October 2022, the owner took goods for his son as a birthday gift. The cost price of the goods was R15 000

Study smarter with the SolutionInn App


