The following reaction involves a conjugate addition reaction followed by an intra molecular Claisen condensation. Write both
Question:
The following reaction involves a conjugate addition reaction followed by an intra molecular Claisen condensation. Write both steps, and show theirmechanisms.
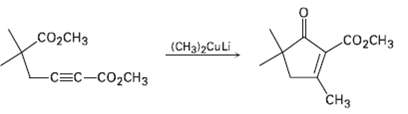
Transcribed Image Text:
сооснз CO2CH3 (CH3i>CuLi -CEC-CO2CH3 сHз
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (15 reviews)
COCH 3 CCCOCH3 HC conjugate ...View the full answer

Answered By

ZIPPORAH KISIO LUNGI
I have worked on several other sites for more than five years, and I always handle clients work with due diligence and professionalism. Am versed with adequate experience in the fields mentioned above in which have delivered quality papers in research, thesis, essays, blog articles, and so forth.
I have gained extensive experience in assisting students to acquire top grades in biological, business and IT papers. Notwithstanding that, I have 7+ years of experience in corporate world software design and development.
5.00+
194+ Reviews
341+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The following reaction involves an intra molecular Michael reaction followed by an intra molecular aldol reaction. Write both steps, and show theirmechanisms. NaOH Ethanol
-
The following reaction involves an intra molecular aldol reaction followed by a ret ro aldol-like reaction. Write both steps, and show theirmechanisms. O2Et CO2Et Na* "OEt Ethanol
-
The following reaction involves a hydrolysis followed by an intra molecular nucleophilic acyl substitution reaction. Write both steps, and show theirmechanisms. CH CH H30* CHCH CH2CO2H
-
Host A has a file of 49.5Kbytes to send to host C and host B has a file of 70.5Kbytes to send to host D, both using TCP. TCP connection 1 from A to C and TCP connection 2 from B to D share one...
-
Which of the following meet the economic definition of an informative signal? a. A man asks for woman's hand in marriage and gives her a large diamond ring. b. The same man takes his fiancée...
-
Calculate by direct integration the moment of inertia for a thin rod of mass M and length L about an axis located distance d from one end. Confirm that your answer agrees with Table 12.2 when d = 0...
-
What is the maximum interest rate I can afford to pay based on the amount of loan I need and the amortization rate offered by the bank?
-
A worker views leisure and income as goods and has an opportunity to work at an hourly wage of $10 per hour. a. Illustrate the workers opportunity set in a given 24-hour period. b. Suppose the worker...
-
Briac Steel sets aside $5300.00 at the beginning of every six months in a fund to replace equipment. If interest is 9% compounded quarterly, how much will be in the fund after 5 years? Hunan bought a...
-
Consider the following approach for testing whether a classifier A beats another classifier B. Let N be the size of a given data set, pA be the accuracy of classifier A, pB be the accuracy of...
-
The Darzens reaction involves a two-step, base-catalyzed condensation of ethyl chloroacetate with a ketone to yield an epoxy ester. The first step is a carbonyl condensation reaction, and the second...
-
The following reaction involves two successive intra molecular Michael reactions. Write both steps, and show theirmechanisms. Nat "OEt Ethanol
-
From the following data prepare the Standard Cost Card for one unit of the sole product manufactured. The budgeted total overheads for year: The fixed overheads (included in the above figures) are...
-
Gary Tuttle has Citiwide Insurance with 100% coverage after a $25.00 copay on office visits. His services today include an office visit ($62.00), urinalysis with differential ($65.00) and a Treadmill...
-
The Elgin Golf Dutton Golf Merger Elgin Golf Inc. has been in merger talks with Dutton Golf Company for the past six months. After several rounds of negotiations, the offer under discussion is a...
-
f ( x ) = x ^ 3 - 3 x ^ 2 - 2 4 x + 5 6 find all critical numbers
-
Suppose a beam of electrons is aimed at two slits in a slide placed in front of a screen. After a short time, the screen looks like the one at the right. a. What evidence does the picture give that...
-
On January 1, Mitzu Company pays a lump-sum amount of $2,700,000 for land, Building 1, Building 2, and Land Improvements 1. Building 1 has no value and will be demolished. Building 2 will be an...
-
Explain employee empowerment in your own words and write a short policy in which you empower the waitstaff in your full-service restaurant. Be sure to include the reasons that you are empowering the...
-
Assume a simple Keynesian depression economy with a multiplier of 4 and an initial equilibrium income of $3,000. Saving and investment equal $400, and assume full employment income is $4,000. a. What...
-
Use the BornHaber cycle and Table 10.3 to calculate the lattice energy of CaO. (H sub for calcium is 178 kJ/mol; IE 1 and IE 2 for calcium are 590 kJ/mol and 1145 kJ/mol, respectively; EA 1 and EA 2...
-
Predict the major products of the following reactions, including stereochemistry. (a) cyclohexene + KMnO4 / H2O (cold, dilute) (b) cyclohexene + peroxyacetic acid in water (c) cis-pent-2-ene + OsO4/...
-
Show how you would accomplish the following conversions. (a) cis-hex-3-ene to meso-hexane-3,4-diol (b) cis-hex-3-ene to (d,l)-hexane-3,4-diol (c) trans-hex-3-ene to meso-hexane-3,4-diol (d)...
-
Give structures of the alkenes that would give the following products upon ozonolysis-reduction. (a) (b) and CH,CH,CH,_C_H cyclohexanone CH,-CH-_-_CH
-
Suppose you bought a bon with an annual coupon rate of 6.5 percent one year ago for $1,032. The bond sells for $1,020 today. a. Assuming a $1,000 face value, what was your total dollar return on this...
-
During the year 2021, William has a job as an accountant, he earns a salary of $100,000. He has done some cleaning services work on his own (self-employed), where he earned a net income of $50,000....
-
Fixed cost per unit is $7 when 25,000 units are produced and $5 when 35,000 units are produced. What is the total fixed cost when 30,000 units are produced? Group of answer choices $150,000....

Study smarter with the SolutionInn App


