Starting with cyclohexene and using any other reagents of your choice, show how you would prepare each
Question:
a.
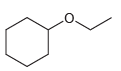
b.
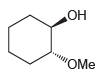
c.
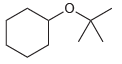
Transcribed Image Text:
OH OMe
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 62% (8 reviews)
a b c...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Starting with cyclohexene and using any other needed reagents, outline a synthesis of 7, 7-dibromobicyclo [4.1.0] heptane.
-
Starting with ethyl acetoacetate, and using any other reagents of your choice, show how you would prepare each of the following compounds. (a) (b) (c) OCH3 Ph.
-
Starting with potassium phthalimide as your source of nitrogen and using any other reagents of your choice, show how you would prepare each of the compounds in Problem 23.18. Compounds in 23.18 (a)...
-
Climate-change concerns are resulting in investment in fuels that dont produce greenhouse gases. Although solar and wind power garner a lot of attention, nuclear power from uranium is also a...
-
Revolve Company's income statement information for 2016 and 2017 (a sole proprietorship) is as follows: Required: Compute the missing amounts labeled (a) through (f). All the necessary information is...
-
One accelerated depreciation method is called fixed-percentage-of-declining-balance. Explain what is meant by the terms "fixed-percentage" and "declining-balance." For what purpose is this method...
-
Question: The Big Red Traveling (BRT) Carnival is in town. Tony arrives at 8:00 p.m., parks in the lot and is robbed at gunpoint by a man who beats him and escapes with his money. There are several...
-
On January 1, 2014, the ledger of Shumway Company contains the following liability accounts. Accounts Payable ........... $52,000 Sales Taxes Payable ........... 5,800 Unearned Service Revenue...
-
thanks . Assume the zero-coupon yields on default-free securities are as summarized in the following table: Maturity 1 year 2 years 3 years 4 years 5 years Zero-Coupon Welds 3.90% 4.20% 4.50% 4.80%...
-
Victoria Rivera owns and manages a consulting firm called Prisek, which began operations on July 1. On July 31, the companys records show the following selected accounts and amounts for the month of...
-
Draw all constitutionally isomeric ethers with molecular formula C 4 H 10 O. Provide a common name and a systematic name for each isomer.
-
When 1, 4-dioxane is heated in the presence of HI, compound A is obtained: a. Draw the structure of compound A. b. If one mole of dioxane is used, how many moles of compound A are formed? c. Show a...
-
Seventy-two percent of Americans squeeze their toothpaste tube from the top. This and other not-so-serious findings are included in The First Really Important Survey of American Habits. Those results...
-
What specific customer needs might employees of a small business have to meet? Explain. How does the changing demographic environment impact the ability of small businesses to compete for customers?...
-
To increase any number by a percent, we can find the growth factor and multiply the number by that growth factor. Find the growth factor when 900 is increased by 16%. The growth factor is
-
1. Discuss why ethics is important in business 2. Refute the businessman's myth that "ethics has no place at all in business." 3. Explain the phrase: "not all that is legal is moral, but what is...
-
20. For the following reaction mechanism use the diagram below to answer each question (this is worth (6 marks) a. The overall reaction is endothermic or exothermic (circle one). b. The 2nd step in...
-
Your assignment is to take a list of strings representing (Department code, course number, title, enrollment) and print them in nice tables, one per line. Data looks like 'CS152Introduction to...
-
Randomly selecting a consumer from California Randomly selecting a consumer who owns a television For each given pair of events, classify the two events as independent or dependent. (If two events...
-
As water moves through the hydrologic cycle, water quality changes are common because of natural phenomena or anthropogenic pollution. Using Figure 11.1, describe how water-quality changes occur...
-
Why arent the glycolysis and gluconeogenesis pathways the exact reverse of one another?
-
Lactate, a product of glucose catabolism in oxygen-starved muscles, can be converted into pyruvate by oxidation. What coenzyme do you think is needed? Write the equation in the normal biochemical...
-
How many moles of acetyl CoA are produced by catabolism of the following substances? (a) 1.0 mol glucose (b) 1.0 mol palmitic acid (c) 1.0 mol maltose
-
Discounts and allowances are effective incentives used primarily to: a. maintain or increase inventory levels in the distribution channel. b. provide special-purpose training to employees. c. reduce...
-
Cash flows from acquiring and selling products are classified as financing activities. operating activities. distribution activities. investing activities
-
In Berg v. Traylor , the court found that: a. Craig could not disaffirm the contract to secure personal management services because it was a contract to provide his and his familys necessaries. b. a...

Study smarter with the SolutionInn App


