The following hypothetical compound cannot be prepared or isolated, because it has a very reactive nucleophilic center
Question:
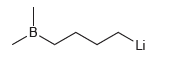
Transcribed Image Text:
B. Li
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 58% (12 reviews)
Electroph...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The following compound cannot be prepared from an alkyl halide or a carboxylic acid using the methods described in this section. Explain why each synthesis cannot be performed. `NH2
-
When atoms of the hypothetical element X are placed together, they rapidly undergo reaction to form the X2 molecule: X(g) + X(g) X2(g) a. Would you predict that this reaction is exothermic or...
-
The hypothetical ionic compound AB2 is very soluble in water. Another hypothetical ionic compound, CB2, is only slightly soluble in water. The lattice energies for these compounds are about the same....
-
Use the limit definition of the definite integral with right Riemann sums and a regular partion to evaluate the following definite integrals. Use the Fundamental Theorem of Calculus to check your...
-
Q1) using the project life cycle develop a matrix showing when various sources of conflict occur. Q2) Describe the communications process in place on your project or in your organization. Q3) List...
-
The purchasing process in a business can be much more complex than most consumer purchasing processes. In about 100 words, outline the differences between a typical consumer purchase and a business...
-
Explain the terms underabsorption and overabsorption.
-
Watters Umbrella Corp. issued 12-year bonds two years ago at a coupon rate of 7.8percent. The bonds make semiannual payments. I these bonds currently sell for 105 percent of par value, what is the...
-
The following data is provided for Garcon Company and Pepper Company Beginning finished goods inventory Beginning work in process inventory Beginning raw materials inventory Rental cost on factory...
-
The balance sheet items for Collier Butcher Shop (arranged in alphabetical order) were as follows at July 1, 2015. (You are to compute the missing figure for Retained Earnings.) During the next few...
-
Identify all of the electrophilic centers in each of the following compounds: a. b. c. Arachidonic acid A precursor in the biosynthesis of many hormones 2-Heptanone Used to control the population of...
-
Each of the following compounds exhibits two electrophilic centers. Identify both centers in each compound. (You will need to draw resonance structures in each case.) a. b. c. H. A cockroach...
-
What Is Intrepreneurship? (pp. 275276)
-
1 Evaluate the given limits using the 10. f(x) = (x-3)(x-5) (a) lim f(x) X-3 (b) lim f(x) x-3+ (c) lim f(x) X-3 graph of the function (d) lim f(x) x+5= (e) lim f(x) x+5+ (f) lim f(x) X-5 50 y -50 2 6...
-
In Year 1 , Stallman Co . had a break - even point of 8 0 , 0 0 0 units, a selling price of $ 1 9 per unit, and fixed costs of $ 2 0 0 , 0 0 0 . What is Stallman Co . s variable cost per unit? (...
-
What will be the output of following statements: int a = 3; if(a!= 3){ cout
-
28. What will this program print, assuming dynamic scoping? (10 points) Consider the following code snippet for problems #29-31. For this problem, assume dynamic scoping. program main() 1. 2. { 3. 4....
-
The reaction X + Y products was studied using the method of initial rates. The initial rate of consumption of X was measured in three different experiments. Data are provided below. Expt [X] 0 (in...
-
In Exercises 7792, use the graph to determine a. The functions domain; b. The functions range; c. The x-intercepts, if any;d. The y-intercept, if any; e. The missing function values, indicated by...
-
Bobbie Singh provides writing services for small businesses. He blogs for companies that need professionally written content. His business records at November 15, 2023, are shown below: During the...
-
Outline a reaction sequence for synthesis of each of the following compounds from the indicated starting material and any other organic or inorganic reagents needed. (a) (b) (c) (d) (e) (f) (g) O C...
-
Linalool, a fragrant compound that can be isolated from a variety of plants, is 3,7-dimethyl-1,6-octadien-3-ol. Linalool is used in making perfumes, and it can be synthesized in the following way:...
-
Compound J, a compound with two four-membered rings, has been synthesized by the following route. Outline the steps that are involved. + Br Et O C (1) LIAIH (2) H20 EtO OEt CsHBr 2 NaOEt (1) HO, HO...
-
Dr. Claudia Gomez, a plastic surgeon, had just returned from a conference in which she learned of a new surgical procedure for removing wrinkles around eyes, reducing the time to perform the normal...
-
QUESTION 2 ( 2 0 Marks ) 2 . 1 REQUIRED Study the information provided below and prepare the Income Statement for the year ended 3 1 December 2 0 2 3 using the marginal costing method. INFORMATION...
-
DROP DOWN OPTIONS: FIRST SECOND THIRD FOURTH 5. Cost of new common stock A firm needs to take flotation costs into account when it is raising capital fromY True or False: The following statement...

Study smarter with the SolutionInn App


