The OH group on the side chain of serine is not deprotonated at a pH of 12.
Question:
Transcribed Image Text:
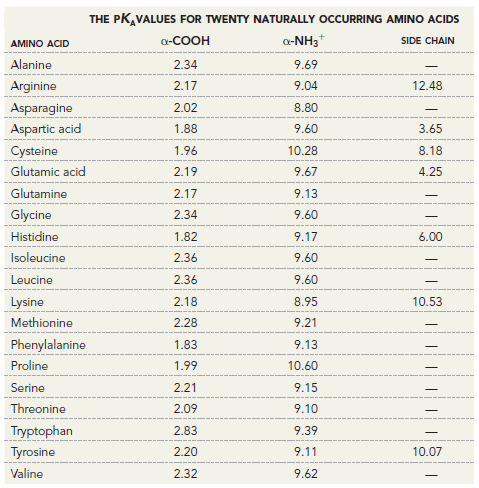
THE PK,VALUES FOR TWENTY NATURALLY OCCURRING AMINO ACIDS a-NH3* a-COOH SIDE CHAIN AMINO ACID Alanine 2.34 9.69 Arginine 2.17 9.04 12.48 Asparagine 2.02 8.80 Aspartic acid 1.88 9.60 3.65 Cysteine 1.96 10.28 8.18 Glutamic acid 9.67 2.19 4.25 Glutamine 9.13 2.17 Glycine 2.34 9.60 Histidine 9.17 1.82 6.00 Isoleucine 2.36 9.60 2.36 9.60 Leucine 8.95 Lysine 2.18 10.53 Methionine 9.21 2.28 Phenylalanine 1.83 9.13 Proline 10.60 1.99 Serine 2.21 9.15 Threonine 2.09 9.10 Tryptophan 2.83 9.39 Tyrosine 2.20 9.11 10.07 Valine 2.32 9.62 T|||||||
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 84% (13 reviews)
Tyrosine possesses a phenolic proton which is more readily depro...View the full answer

Answered By

Jane Thuita
I am a highly motivated and experienced tutor with a passion for helping students succeed. I have a strong background in a variety of academic subjects, including mathematics, science, and English. I have experience working with students of all ages and abilities, and I am able to customize my teaching methods to best fit each student's unique learning style. I am also able to work with students who have learning disabilities and those who are English language learners. I am patient, understanding, and dedicated to helping my students reach their full potential. In my free time, I enjoy reading, playing sports, and spending time with my family. I am excited to work with you and help you achieve your goals.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The citric acid cycle is a series of biological reactions that plays a central role in cell metabolism. The cycle includes dehydration reactions of both malic and citric acids, yielding fumaric and...
-
There are a number of conserved sequences found in an mRNA which dictate where splicing occurs. Where are these sequences found relative to the exon/intron junctions? What is the significance of...
-
Histidine possesses a basic side chain which is protonated at physiological pH. Identify which nitrogen atom in the side chain is protonated.
-
Once down to about 15, the worlds only wild flock of whooping cranes now numbers a record 237 birds in its Texas Coastal Bend wintering ground (www.SunHerald.com). The average whooping crane egg...
-
Marcel Company projects the following sales for the first three months of the year: $11,200 in January; $12,300 in February; and $11,100 in March. The company expects 60% of the sales to be cash and...
-
What is the difference between the local truncation error and the global error, and how are they related?
-
The cost unit for water supplies is (a) per 1,000 gallons (b) per lorry (c) per dozen litres (d) per consumer
-
Blanda Incorporated management is considering investing in two alternative production systems. The systems are mutually exclusive, and the cost of the new equipment and the resulting cash flows are...
-
Assessment 2: Descriptive Analysis, Prediction and Communication Task overview In this assessment, students will be provided a business scenario and dataset. Students will be required to use the...
-
2. Azali Ltd is a large fashion retailer that has operations in many countries. The company wants to raise more funds for its operations. For this purpose the directors have decided to make a...
-
Using the data in the following table, calculate the pI of the following amino acids. (a) Aspartic acid (b) Leucine (c) Lysine (d) Proline THE PK,VALUES FOR TWENTY NATURALLY OCCURRING AMINO ACIDS...
-
Explain why it is inappropriate to use a chiral catalyst in the preparation of glycine.
-
Give all of the product(s) expected, including pertinent stereochemistry, when each of the following compounds reacts with sodium ethoxide in ethanol. (D = deuterium = 2H, an isotope of hydrogen.)...
-
Given the following memory status below, compute how much does it cost to compact holes together with the following compaction strategies if 1 kbyte of movement costs 50 centavos. 0. OS OS OS OS OS...
-
ITG Pte Ltd ("ITG") is a company specialising in air-conditioner maintenance and servicing. It makes adjusting and closing entries every 31 December, which is the company's financial year-end. Unless...
-
(20 points) We know that when we have a graph with negative edge costs, Dijkstra's algo rithm is not guaranteed to work. (a) Does Dijkstra's algorithm ever work when some of the edge costs are...
-
Create a new user called cis605_usr. Use Master. assign a password of abcd, set check_policy to off and check_expiration to off (Why set these two to off?). Execute the sp_addsrvrolemember to add the...
-
Salmone Company reported the following purchases and sales of its only product. Salmone uses a periodic inventory system. Determine the cost assigned to the ending inventory using FIFO. Date Units...
-
Gull Corporation, a cash method, calendar year C corporation, was formed and began business on November 1, 2021. Gull incurred the following expenses during its first year of operations (November 1,...
-
a. What is meant by the term tax haven? b. What are the desired characteristics for a country if it expects to be used as a tax haven? c. What are the advantages leading an MNE to use a tax haven...
-
In the addition of just 1 mole of bromine to 1 mole of hex-1-yne, should the hex-1-yne be added to a bromine solution or should the bromine be added to the hex-1-yne? Explain your answer.
-
Propose a mechanism for the entire reaction of pent-1-yne with 2 moles of HBr. Show why Markovnikov's rule should be observed in both the first and second additions of HBr.
-
Predict the major product(s) of the following reactions: (a) phenylacetylene + 2 HBr (b) hex-1-yne + 2 HBr (c) cyclooctyne + 2 HCl (d) hex-2-yne + 2 HCl + 2 HBr
-
Docs Auto Body has budgeted the costs of the following repair time and parts activities for 2009: Doc's budgets 6,000 hours of repair time in 2009. A profit margin of $7 per labour hour will be added...
-
QUESTION 28 In a perpetual inventory system, the cost of inventory sold is: Debited to accounts receivable. Debited to cost of goods sold. O Not recorded at the time goods are sold. O Credited to...
-
The following financial statements and additional information are reported. IKIBAN INC. Comparative Balance Sheets June 30, 2019 and 2018 2019 2018 $105,709 69,500 66,800 4,700 246,700 127,eee...

Study smarter with the SolutionInn App


