A box like the one in Problem 31 has two particles in compartment A, two particles in
Question:
A box like the one in Problem 31 has two particles in compartment A, two particles in compartment B, and four energy units to be distributed.
(a) Construct a table like Table 19. 2 (page 656) for this setup.
(b) Does equipartition of energy take place even with such a small number of particles?
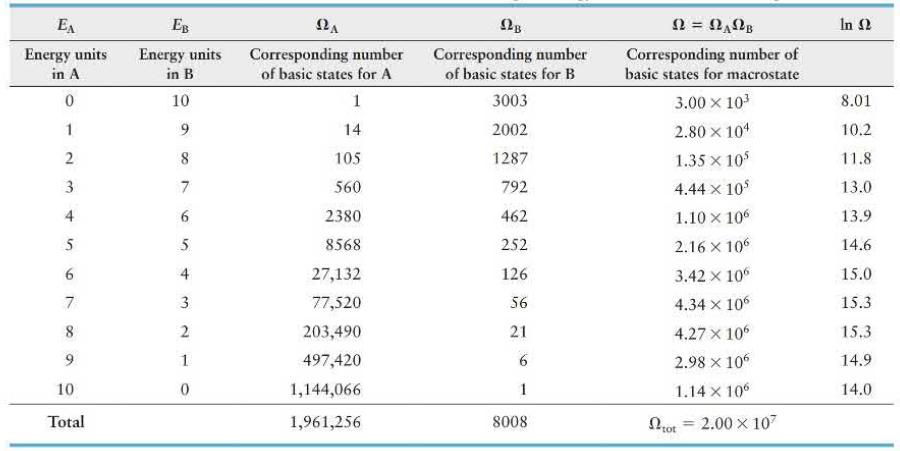
Data from Problem 31
A box divided into compartments \(A\) and \(B\) contains 14 particles in \(A\) and 6 in \(B\). The separating partition allows energy exchange between compartments as the particles collide with the partition. Table 19. 2 (page 656) lists the numbers of basic states possible when 10 energy units are distributed in various combinations over the 20 particles. How much more likely is it for compartment \(A\) to contain 7 energy units (macrostate \(E_{\mathrm{A}}=7, E_{\mathrm{B}}=3\) ) than for this compartment to contain 3 energy units (macrostate \(E_{\mathrm{A}}=3, E_{\mathrm{B}}=7\) )?
Step by Step Answer:






