Conductivity measurements were one of the first methods used to determine the autoionization constant of water. The
Question:
![[н] (он ] ад аон 1м K. 1м](https://dsd5zvtm8ll6.cloudfront.net/si.question.images/images/question_images/1526/3/6/6/6755afa81d3011be1526366669622.jpg)
where a is the activity of the species, which is equal to the actual concentration of the species divided by the standard state concentration at infinite dilution. This substitution of concentrations for activities is a reasonable approximation
given the small concentrations of H+ and OHˆ’ that result from autoionization.
a. Using the expression provided, show that the conductivity of pure water can be written as
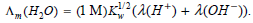
b. Kohlrausch and Heydweiller measured the conductivity of water in 1894 and determined that Λm (H2O) = 5.5 × 10-6 S m-1 at 298 K. Using the information provided in Table 34.2, determine Kw.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

Question Posted:





