Consider the comparison made between accurate results and those based on calculations using the van der Waals
Question:
Figure 7.1
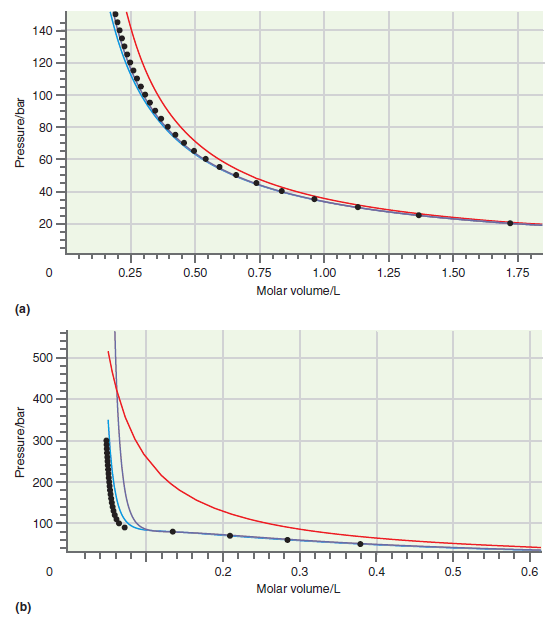
Figure 7.5
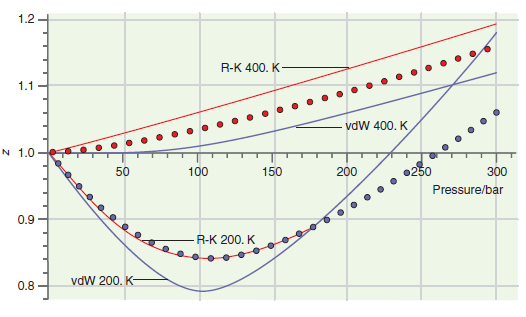
Transcribed Image Text:
140 120 100 80 40 20 0.25 0.75 0.50 1.00 1.25 1.50 1.75 Molar volume/L (a) 500 400 300 200 100 0.2 0.3 0.4 0.5 0.6 Molar volume/L (b) Pressure/bar Pressure/bar 1.2 R-K 400. K 1.1 vdW 400. K N 1.0 100 150 200 •250 300 Pressure/bar 0.9 R-K 200. K vdW 200. K- 0.8
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (15 reviews)
The RedlichKwong gives more ac...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Explain why the oscillations in the two-phase coexistence region using the RedlichKwong and van der Waals equations of state (see Figure 7.4) do not correspond to reality. Figure 7.4 140 120 100 304...
-
The van der Waals equation of state, an approximate representation of the behavior of gases at high pressure, is given by Where a and b are constants having different values for different gases. (In...
-
Derive the following relation, for the internal pressure of a gas that obeys the RedlichKwong equation of state, aU av /T 2TV, + b) || RT 1 V , + b)
-
1. As shown by point D in Fig 3.1, the volume of an ideal diatomic gas is 2.00L at standard condition (STP, T=273.15K, P=101.3kPa). The gas is heated to A with its volume conserved, expands...
-
Select a region of the world. Research current events and discuss how economic, political, and social changes will impact growth in the region. Write a short paper summarizing your findings.
-
Find both first partial derivatives. (x, y) = 2x - 5y + 3
-
4. If a bank increases its maturity mismatch, what happens to its economic spread before taxes and its economic spread after taxes (i.e., including the tax penalty)?
-
Fred was the owner of a three-bedroom cabin in California. During 2013, he contracted with a property-management company to rent the cabin to third parties. In exchange for its services, Fred paid...
-
Which of the following is not considered a capital budgeting project? Purchase of a new packaging machine Purchase of land on which to build a new factory Purchase of a new delivery truck to replace...
-
According to an article in USA Today, 90% of Americans will suffer from high blood pressure as they age. Out of 20 randomly chosen people what is the probability that at most 3 will suffer from high...
-
We return to the 60. kg hiker of P4.34, who is climbing the 828 m tall Burj Khalifa in Dubai. If the efficiency of converting the energy content of the bars into the work of climbing is 25%, the...
-
The initiation step for radical addition of HBr is highly endothermic: (a) Explain how this step can be thermodynamically favorable at high temperature even though it is endothermic. (b) Explain why...
-
You will earn the YTM on a bond if you hold the bond until maturity and if interest rates dont change. If you actually sell the bond before it matures, your realized return is known as the holding...
-
Investigate the Mercedes Benz company and you have to cover this topic " For Business prospects, Market growth, Market quality, and Environmental aspects are three most important factors. Explain...
-
The case study for Goodwill Industries and how they "do good" as a core business strategy. What are Goodwill's competitive advantages? Goodwill has found success in social services. What problems...
-
Cosmic Cals (Pty) Ltd , a seller of personalized scientific calculators, had an inventory of 40 calculators. The value of these calculators is R15 400 each on the 1 January 2022. During the current...
-
Perform an analysis of Best Buy Co. Inc. Your analysis will draw on the Form 10K (as of February 2013). Your analysis can include information prior to February 2013 but should not draw on any...
-
Research organizational structure of a company of your choice. Use your understanding of organizational structure to analyze whether this organization's structure is the best choice for the business...
-
Let (x) = x 2 - 9, g(x) = 2x, and h(x) = x - 3. Find each of the following. (7) (x)
-
2.) Find the Laplace transform of f(t) 7e-St cos 2t +9 sinh2 2t. Use Laplace Table. %3D
-
Suppose that a molecular orbital has the form N (O.l45A + 0.844B). Find a linear combination of the orbitals A and B that is orthogonal to this combination.
-
Show that, if a wave cos kx centred on A (so that x is measured from A) interferes with a similar wave cos k' centred on B (with x measured from B) a distance R away, then constructive interference...
-
Before doing the calculation below, sketch how the overlap between a is orbital and a 2p orbital can be expected to depend on their separation. The overlap integral between an H Is orbital and an H2p...
-
Los datos de la columna C tienen caracteres no imprimibles antes y despus de los datos contenidos en cada celda. En la celda G2, ingrese una frmula para eliminar cualquier carcter no imprimible de la...
-
Explain impacts of changing FIFO method to weighted average method in inventory cost valuations? Explain impacts of changing Weighted average method to FIFO method in inventory cost valuations?...
-
A perpetuity makes payments starting five years from today. The first payment is 1000 and each payment thereafter increases by k (in %) (which is less than the effective annual interest rate) per...

Study smarter with the SolutionInn App


