Identify the molecular orbitals for F 2 in the images shown here in terms of the two
Question:
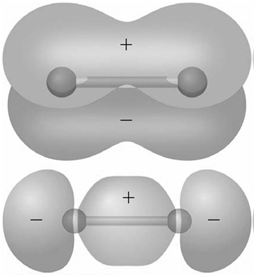
Transcribed Image Text:
+
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 45% (11 reviews)
The top image is of symmetry and has no ...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
(a) Draw the molecular orbitals for the cyclopropenyl case. (Since there are three p orbitals, there must be three MOs: one all-bonding MO and one degenerate pair of MOs.) (b) Draw an energy diagram...
-
Evaluate the Klopp case in terms of those two criteria.
-
Images of molecular orbitals for LiH calculated using the minimal basis set are shown here. In these images, the smaller atom is H. The H1s AO has a lower energy than the Li2s AO. The energy of the...
-
9:49 X Bank Reconciliation Assignment 1 PDF 150 KB Assignment #1 - Bank Reconciliation This assignment has 33 total marks. This assignment is worth 5% of your final grade. This assignment is due...
-
A police car with its spotlight on is parked 20 meters from a warehouse. Consider and x as shown in the figure. (a) Write θ as a function of x. (b) Find θ when x = 5 meters...
-
Choose the best word in the sentences below. We needed to (adapt/adopt/adept) the play to make it appropriate for younger audiences.
-
Sports news on local TV broadcasts. The Sports Journal LO9 (Winter 2004) published the results of a study conducted to assess the factors that affect the time allotted to sports news on local...
-
The times required to complete each of eight jobs in a two-machine flow shop are shown in the table that follows. Each job must follow the same sequence, beginning with machine A and moving to...
-
how to find the missing Molly Spencer, capital? In this project, you will use skills and procedures presented in Chapters 1-3 together as you complete journal entries and financial statements for NJW...
-
This case study on project evaluation is applicable for beginning courses in corporate finance or finance strategy. Two alternative investment options are available to evaluate. Challenges are...
-
If there is a node in u , is the electron in this wave function really delocalized? How does it get from one side of the node to the other?
-
By considering each term in And Explain why the values of J and K are positive for H + 2 . e? |e, dr - j (, Hs, dt J = [@n| dt 4TEQ7B.
-
Income Statement and Balance Sheet Green Bay Corporation began business in July 2008 as a commercial fishing operation and a passenger service between islands. Shares of stock were issued to the...
-
Share your thoughts on the descriptions of coaching versus mentoring. Discuss which technique you personally find more helpful, incorporating your peers' example scenarios if possible. Provide...
-
Hanung Corp has two service departments, Maintenance and Personnel. Maintenance Department costs of $380,000 are allocated on the basis of budgeted maintenance-hours. Personnel Department costs of...
-
Discuss difference between nominal interest rate and real interest rate. Explain why real interest rate is more important than the nominal interest rate using your answer to Question 1 of the...
-
Refer to Figure 14-1. How would an increase in the money supply move the economy in the short and long run?
-
1) Special Relativity. Statement: Imagine this situation: Alice stands in New York City while Bob, aboard a plane departing from Boston, directly crosses over Alice at t=0. Disregard the vertical...
-
In Exercises 5358, f and g are defined by the following tables. Use the tables to evaluate each composite function. f -1 (g(1)) X -1 0 1 2 f(x) 1 4 5 -1 X -1 1 4 10 g(x) 0 1 2 -1
-
a. What is meant by the term tax haven? b. What are the desired characteristics for a country if it expects to be used as a tax haven? c. What are the advantages leading an MNE to use a tax haven...
-
The CH 3 Cl molecule belongs to the point group C 3v . List the symmetry elements of the group and locate them in the molecule.
-
Show that, for a perfect gas, (U/S) V = T and (U/V) S = p.
-
Sketch the form of the 19 F-NMR spectra of a natural sample of tetrafluoroborate ions, BF 4 , allowing for the relative abundances of 10 BF 4 and 11 BF 4 .
-
Famas Llamas has a weighted average cost of capital of 8.8 percent. The companys cost of equity is 12 percent, and its pretax cost of debt is 6.8 percent. The tax rate is 22 percent. What is the...
-
The common stock of a company paid 1.32 in dividens last year. Dividens are expected to gros at an 8 percent annual rate for an indefinite number of years. A) If the company's current market price is...
-
(1 point) Bill makes annual deposits of $1900 to an an IRA earning 5% compounded annually for 14 years. At the end of the 14 years Bil retires. a) What was the value of his IRA at the end of 14...

Study smarter with the SolutionInn App


