The vapor pressure of ethanol(l) is given by a. Calculate the standard boiling temperature. b. Calculate ÎH
Question:
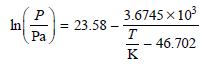
a. Calculate the standard boiling temperature.
b. Calculate ΔH vaporization at 298 K and at the standard boiling temperature.
Transcribed Image Text:
3.6745 x 10 23.58 In Pa – - 46.702 K
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 61% (13 reviews)
a In b Pa 20767 ...View the full answer

Answered By

Hamza Amjad
Currently I am student in master degree program.from last two year I am tutring in Academy and I tought many O/A level student in home tution.
4.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The vapor pressure of an unknown solid is approximately given by ln(P/Torr) = 22.413 2211 (K/T), and the vapor pressure of the liquid phase of the same substance is approximately given by ln(P/Torr)...
-
Carbon tetrachloride melts at 250. K. The vapor pressure of the liquid is 10,539 Pa at 290. K and 74,518 Pa at 340. K. The vapor pressure of the solid is 270. Pa at 232 K and 1092 Pa at 250. K. a....
-
Consider the process which is carried out at constant pressure. The total DS for this process is known to be 75.0 J K-1mol-1. For A(l) and A(g), the Cp values are 75.0 JK-1mol-1 and 29.0 JK-1mol-1,...
-
8 for 0 < < 6 for 6
-
Discus the Ten Strategies of a World-Class Computer Security Incident Response Team, Research and find at least three (3) more recommendations needed to organize, fund and introduce a CSIRT. Research...
-
Find potential functions for the field. F = 2i + (2y + z)j + (y + 1)k
-
What are we trying to do?
-
Babbit, Inc., a multinational corporation based in the United States, owns an 80% interest in Nakima Company, which is located in Sydney, Australia. The acquisition occurred on January 1, 2008. The...
-
Question 22 Yaeko Falck wit receive an annuity of 57 270.00 avery four monta for 21 years. How much is this how worth to the tota 5366,574 54 $220.06409 51.158,316.14 572,802.10 Question as 4116...
-
Given IDSS = 9 mA and VP = -3.5 V, determine ID when: a. VGS = 0V. b. VGS = -2V c. VGS = -3.5V d. VGS = -5V
-
Use the vapor pressures of tetrachloromethane given in the following table to calculate the enthalpy of vaporization using a graphical method or a least squares fitting routine. P/Pa T (K) 320. 330....
-
In Section 8.8, it is stated that the maximum height of a water column in which cavitations does not occur is 9.7 m. Show that this is the case at 298 K.
-
In Problems 5968, express each sum using summation notation. 1+ 3 + 5 + 7 + + [2(12) - 1]
-
As an official sponsor of the Olympics, what specific benefit did John Hancock use to help drive sales in their national offices?
-
assumes that Nia has both a discount rate of zero and faces an interest rate of zero. These assumptions made calculating her constant level of consumption expenditure of $56,000 fairly...
-
Paul Petersen lives in Northern California. He owns a BMW car worth about $20,000. He wants to take a trip to Nevada with his girlfriend Patricia, who lives in Los Angeles. He takes his car into...
-
Do you see gendered patterns of interaction in personal relationships? Does knowing about gender linked patterns affect how other interpret on what happens in a relationships?
-
Significance For bone density scores that are normally distributed with a mean of 0 and a standard deviation of 1, find the percentage of scores that are significantly high (or at least 2 standard...
-
Experts fear that without conservation efforts, tigers could disappear from the wild by 2022. Just one hundred years ago, there were at least 100,000 wild tigers. By 2010, the estimated world tiger...
-
Bobbie Singh provides writing services for small businesses. He blogs for companies that need professionally written content. His business records at November 15, 2023, are shown below: During the...
-
Find the 3 by 3 matrix that is equivalent in its action to each of the symmetry operators: (a) S 2(z) . (b) C 2(x) .
-
Find S 2(y) (3,4,5).
-
Find the result of each operation on the given point (represented by Cartesian coordinates): (a) C 2(z) i (1,1,1). (b) i C 2(z) (1,1,1).
-
Create a Data Table to depict the future value when you vary the interest rate and the investment amount. Use the following assumptions: Interest Rates: Investment Amounts:-10.0% $10,000.00 -8.0%...
-
Isaac earns a base salary of $1250 per month and a graduated commission of 0.4% on the first $100,000 of sales, and 0.5% on sales over $100,000. Last month, Isaac's gross salary was $2025. What were...
-
Calculate the price, including both GST and PST, that an individual will pay for a car sold for $26,995.00 in Manitoba. (Assume GST = 5% and PST = 8%) a$29,154.60 b$30,234.40 c$30,504.35 d$28,334.75...

Study smarter with the SolutionInn App


