Use the vapor pressures of tetrachloromethane given in the following table to calculate the enthalpy of vaporization
Question:
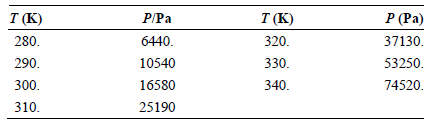
Transcribed Image Text:
P/Pa T (K) 320. 330. P (Pa) 37130. T (K) 280. 6440. 53250. 74520. 290. 10540 300. 16580 340. 25190 310.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (12 reviews)
A least squares fit of ln P ver...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Use the vapor pressures for tetrachloromethane given in the following table to estimate the temperature and pressure of the triple point and also the enthalpies of fusion, vaporization, and...
-
Use the values for nominal GDP and real GDP given in the following table to calculate the inflation rate during 1930: 1929 5103.6 billion 5977.0 billion 1930 Nominal GDP Real GDP $91.2 billion $892.8...
-
Use the vapor pressures of ice given here to calculate the enthalpy of sublimation using a graphical method or a least squares fitting routine. T (K) P (Torr) 200. 0.1676 210. 0.7233 2.732 220. 230....
-
The JoFe Computers and Accessory Company produces two types of laptop computer bags. Version A costs $32, takes 4 hours of labor, and sells for $50. Version B costs $38, takes 6 hours of labor, and...
-
1). Critically analyze current European and United States industry standards or recommendations for any Information Technology (IT) area or subarea (e.g., intrusion detection, data recovery, data...
-
Use the curl integral in Stokes Theorem to find the circulation of the field F around the curve C in the indicated direction. C: The ellipse in which the plane 2x + 6y - 3z = 6 meets the cylinder x 2...
-
introduce basic techniques for process mapping and suggest ways in which process changes might draw on such modelling approaches Use the following questions to challenge the assumptions that underpin...
-
In her book Seven Signs of Ethical Collapse, Jennings explains: When an organization collapses ethically, it means that those in the organization have drifted into rationalizations and legalisms, and...
-
Give two uses of the Islamic stock market indices. OR Give two reasons why Islamic stock market indices were developed
-
In the 2009?2010 academic year, many public universities in the United States raised tuition and fees due to a decrease in state subsidies. The change in the cost of tuition, a shared dormitory room,...
-
20.0 g of water is in a container of 20.0 L at 298.15 K. The vapor pressure of water at this temperature is 23.76 Torr. a. What phases are present? b. At what volume would only the gas phase be...
-
The vapor pressure of ethanol(l) is given by a. Calculate the standard boiling temperature. b. Calculate ÎH vaporization at 298 K and at the standard boiling temperature. 3.6745 x 10 23.58 In...
-
Which of the following is NOT an advantage of object-oriented approaches? (a) Low maintenance costs (b) Follows the life-cycle model sequentially (c) Seamless progression between development stages...
-
do you agree wih this approach to dismantling the toxic culture? explain
-
Movies When randomly selecting a speaking character in a movie, the probability of getting a female is 0.331 (based on data from "Inequality in 1200 Popular Films," by Smith, et al., Annenberg...
-
Steve Reese is a well-known interior designer in Fort Worth, Texas. He wants to start his own business and convinces Rob O'Donnell, a local merchant, to contribute the capital to form a partnership....
-
Exercise 6-10A (Algo) Double-declining-balance and units-of-production depreciation: gain or loss on disposal LO 6-3, 6-4, 6-5 Exact Photo Service purchased a new color printer at the beginning of...
-
Independent Events Again assume that when randomly selecting a speaking character in a movie, the probability of getting a female is 0.331, as in Exercise 1. If we want to find the probability of 20...
-
In Exercises 7374, use the graph of the rational function to solve each inequality. f(x) = x + 1 1-4 [-4, 4, 1] by [-4, 4, 1]
-
The activities listed in lines 2125 serve primarily as examples of A) Underappreciated dangers B) Intolerable risks C) Medical priorities D) Policy failures
-
List the symmetry elements for (a) H 2 O (bent). (b) CO 2 (linear).
-
Find the 3 by 3 matrix that is equivalent in its action to each of the symmetry operators: (a) C 8(x) . (b) S 6(x) .
-
List the symmetry elements of a uniform cube centered at the origin with its faces perpendicular to the coordinate axes.
-
Fig 1. Rolling a 4 on a D4 A four sided die (D4), shaped like a pyramid (or tetrahedron), has 4 flat surfaces opposite four corner points. A number (1, 2, 3, or 4) appears close to the edge of each...
-
I just need help with question #4 please! Thank you! Windsor Manufacturing uses MRP to schedule its production. Below is the Bill of Material (BOM) for Product A. The quantity needed of the part...
-
(25) Suppose that we have an economy consisting of two farmers, Cornelius and Wheaton, who unsurprisingly farm corn c and wheat w, respectively. Assume that both farmers produce their crop of choice...

Study smarter with the SolutionInn App


