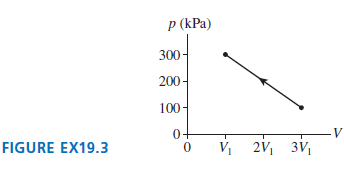
80 J of work are done on the gas in the process shown in Figure EX19.3. What
Question:
Transcribed Image Text:
p (kPa) 300- 200- 100- VI 2V1 FIGURE EX19.3 3V1
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (6 reviews)
Model Assume the gas is ideal The work done on a gas is the negative of the area under the ...View the full answer

Answered By

WAHIDUL HAQUE
hello,
I'm a professional academic solution provider working as a freelance academic solution provider since 7 years. I have completed numerous projects. Help lots of students to get good marks in their exams and quizzes. I can provide any type of academic help to your homework, classwork etc, if you are a student of Accounting, Finance, Economics, Statistics. I believe in satisfying client by my work quality, rather than making one-time profit. I charge reasonable so that we make good long term relationship. why will you choose me? i am an extremely passionate, boldly honest, ethically driven and pro-active contractor that holds each of my clients in high regards throughout all my business relations. in addition, I'll always make sure that I'm giving my 100% better in every work that will be entrusted to me to be able to produce an outcome that will meet my client's standards. so if you are a student that is now reading my profile and considering me for your academic help. please feel free to look through my working history, feedback and contact me if you see or read something that interests you. I appreciate your time and consideration.
regards
4.90+
233+ Reviews
368+ Question Solved
Related Book For 

Physics for Scientists and Engineers A Strategic Approach with Modern Physics
ISBN: 978-0133942651
4th edition
Authors: Randall D. Knight
Question Posted:
Students also viewed these Physics questions
-
Three different processes act on a system. (a) In process A, 42 J of work are done on the system and 77 J of heat are added to the system. Find the change in the system's internal energy. (b) In...
-
500 J of work are done on a system in a process that decreases the systems thermal energy by 200 J. How much heat energy is transferred to or from the system?
-
Juana slides a crate along the floor of the moving van. The coefficient of kinetic friction between the crate and the van floor is 0.120. The crate has a mass of 56.8 kg and Juana pushes with a...
-
Bloomberg Intelligence listed 50 companies to watch in 2018 (www .bloomberg.com/features/companies-to-watch-2018). Twelve of the companies are listed here with their total assets and 12-month sales....
-
The neutrons in a parallel beam, each having kinetic energy 0.025 eV, are directed through two slits 0.40 mm apart. How far apart will the interference peaks be on a screen 1.0 m away?
-
If we do not reject H0, we conclude that H0 is true. In Exercises 912, determine whether the statement is true or false. If the statement is false, rewrite it as a true statement.
-
Kablossom started a business in 2010 with total assets of \($35,000\) and total liabilities of \($32,000\). At the end of 2010, Kablossoms total assets were \($65,000\), and total liabilities were...
-
Are some beer, wine, or spirits ads misleading? What examples can you give? What is misleading in them? Do some ads contain images and themes that go too far in appealing to an audience under the...
-
How can parties that have unequal bargaining power negotiate meaningfully, without one party taking advantage of the other? Have you ever negotiated with someone who had more bargaining power than...
-
A 10-year-old truck had operating costs last year of $3,200, and it is expected A 10 that these costs will increase at the rate of 4% per year if there is no inflation Presently the truck can be sold...
-
Do (a) Temperature, (b) Heat, (c) Thermal energy describe a property of a system, an interaction of the system with its environment, or both? Explain.
-
Two containers hold equal masses of nitrogen gas at equal temperatures. You supply 10 J of heat to container A while not allowing its volume to change, and you supply 10 J of heat to container B...
-
A $25 000, 6% bond with semi-annual coupons redeemable at par in 20 years is purchased to yield 8% compounded semi-annually. Determine the gain or loss if the bond is sold 7 years after the date of...
-
You are generating your pro forma income statement for next year. Sales are forecast to be $100,000. Variable operating expenses are expected to be 20% of sales, and fixed operating expenses are...
-
Assume BBB stock price is currently $20. In each six-month period, it will either fall by 50 percent or rise by 100 percent. What is the current value of a one-year call option with an exercise price...
-
A parked police car takes off after a suspect and reaches 78.5 km/h East in 8.20 seconds. What is the average acceleration (m/s^2) of the car? | Select an answer Enter an integer or decimal number,...
-
1. What is the essence of communication for women? How about for men? 2. Describe the masculinity-femininity dimension and offer a specific example not listed in the reading. 3. Why is there a...
-
How can we manage organizational changes effectively. b) How important are managers in the organizational change process.?
-
Consider a region of space in which the energy density stored in the magnetic field (Eq. 21.35) is equal to the energy density in the electric field (Eq. 18.48). What is the ratio B/E? Express your...
-
Consider the advantages and disadvantages of extending property rights so that everyone would have the right to prevent people imposing any costs on them whatsoever (or charging them to do so).
-
Iodine-131, a beta emitter, has a half-life of 8 days. A 2-gram sample of initially pure iodine-131 is stored for 32 days. How much iodine-131 remains in the sample afterward?
-
The amount of carbon-14 in an ancient wooden bowl is found to be one-half that in a new piece of wood. How old is the bowl?
-
A nucleus of element 112coperniciumis formed using the reaction equation given near the end of Section 11.7. It then undergoes six successive alpha decays. Give the identity of the isotope that...
-
Fanning Corporation estimated its overhead costs would be $23,300 per month except for January when it pays the $132,240 annual insurance premium on the manufacturing facility. Accordingly, the...
-
Restore Company manufactures jeans. In June, Restore made 500 pairs of jeans, but had budgeted production at 800 pairs of jeans. The allocation base for overhead costs is direct labor hours. The...
-
Eevee's Chemicals started operations on Dec 1. Inventory records for Eevee's Chemicals revealed the following transactions during December: Units Unit Cost Total Cost Purchases: Dec. 2 200 $3.00 $600...

Study smarter with the SolutionInn App


