A molecular bond can be modeled as a spring between two atoms that vibrate with simple harmonic
Question:
Transcribed Image Text:
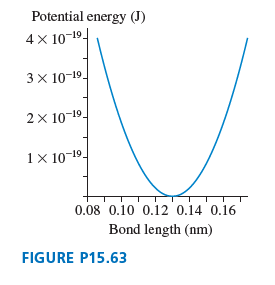
Potential energy (J) 4 x 10-19- 3x 10-19- 2x 10-19- 1 × 10-19- 0.08 0.10 0.12 0.14 0.16 Bond length (nm) FIGURE P15.63
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (12 reviews)
Solve The potential energy curve of a simple harmonic oscillator is des...View the full answer

Answered By

Ashington Waweru
I am a lecturer, research writer and also a qualified financial analyst and accountant. I am qualified and articulate in many disciplines including English, Accounting, Finance, Quantitative spreadsheet analysis, Economics, and Statistics. I am an expert with sixteen years of experience in online industry-related work. I have a master's in business administration and a bachelor’s degree in education, accounting, and economics options.
I am a writer and proofreading expert with sixteen years of experience in online writing, proofreading, and text editing. I have vast knowledge and experience in writing techniques and styles such as APA, ASA, MLA, Chicago, Turabian, IEEE, and many others.
I am also an online blogger and research writer with sixteen years of writing and proofreading articles and reports. I have written many scripts and articles for blogs, and I also specialize in search engine
I have sixteen years of experience in Excel data entry, Excel data analysis, R-studio quantitative analysis, SPSS quantitative analysis, research writing, and proofreading articles and reports. I will deliver the highest quality online and offline Excel, R, SPSS, and other spreadsheet solutions within your operational deadlines. I have also compiled many original Excel quantitative and text spreadsheets which solve client’s problems in my research writing career.
I have extensive enterprise resource planning accounting, financial modeling, financial reporting, and company analysis: customer relationship management, enterprise resource planning, financial accounting projects, and corporate finance.
I am articulate in psychology, engineering, nursing, counseling, project management, accounting, finance, quantitative spreadsheet analysis, statistical and economic analysis, among many other industry fields and academic disciplines. I work to solve problems and provide accurate and credible solutions and research reports in all industries in the global economy.
I have taught and conducted masters and Ph.D. thesis research for specialists in Quantitative finance, Financial Accounting, Actuarial science, Macroeconomics, Microeconomics, Risk Management, Managerial Economics, Engineering Economics, Financial economics, Taxation and many other disciplines including water engineering, psychology, e-commerce, mechanical engineering, leadership and many others.
I have developed many courses on online websites like Teachable and Thinkific. I also developed an accounting reporting automation software project for Utafiti sacco located at ILRI Uthiru Kenya when I was working there in year 2001.
I am a mature, self-motivated worker who delivers high-quality, on-time reports which solve client’s problems accurately.
I have written many academic and professional industry research papers and tutored many clients from college to university undergraduate, master's and Ph.D. students, and corporate professionals. I anticipate your hiring me.
I know I will deliver the highest quality work you will find anywhere to award me your project work. Please note that I am looking for a long-term work relationship with you. I look forward to you delivering the best service to you.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Physics for Scientists and Engineers A Strategic Approach with Modern Physics
ISBN: 978-0133942651
4th edition
Authors: Randall D. Knight
Question Posted:
Related Video
simple harmonic motion, in physics, repetitive movement back and forth through an equilibrium, or central, position, so that the maximum displacement on one side of this position is equal to the maximum displacement on the other side. The time interval of each complete vibration is the same
Students also viewed these Physics questions
-
The bumper on a car is made of flexible plastic so that it will compress on impact and cushion the cars occupants during a collision. Suppose a car of mass m = 1200 kg is traveling slowly through a...
-
A flat roof can be modeled as a flat plate insulated on the bottom and placed in the sunlight. If the radiant heat that the roof receives from the sun is 600 W/m2, the convection heat transfer...
-
A coffee maker can be modeled as a heating element (resistance R) connected to the outlet voltage of 120 V (assumed to be dc). The heating element boils small amounts of water at a time as it brews...
-
Refer to the bolt strength problem 17.47. Assume = 6,050 and 5 100. Use the following 24 individual bolt strength observations to answer the questions posed. (a) Prepare a histogram and/or normal...
-
The trial balance of Brentwood Construction at July 31, 2014, appears below Additional data at July 31, 2014: a. Amortization for the period to be recorded: equipment, $2,040; building, $4,210. b....
-
Task Fifteen Bryans Corporation has provided the following data for its two most recent years of operation: $ 53 Selling price per unit Manufacturing costs: Variable manufacturing cost per unit...
-
The major roadblock to foreign companies listing their overseas stock on U.S. exchanges has long been the big difference between accounting standards in the United States and abroad. Recently,...
-
Mike Polanski is 30 years of age and his salary next year will be $40,000. Mike forecasts that his salary will increase at a steady rate of 5% per annum until his retirement at age 60. a. If the...
-
5 determine the unit product cost of each product according to the ABC system and round your final answer to second decimal place. please help thank you Direct Labor-Hours per unit 0.40 0.70 Rims...
-
If Jane is not maximizing her utility, remembering the Law of Diminishing Marginal Utility, would she be better off to buy one less chicken sandwich and one more fry? Explain your reasoning and show...
-
FIGURE P15.62 is a top view of an object of mass m connected between two stretched rubber bands of length L. The object rests on a frictionless surface. At equilibrium, the tension in each rubber...
-
A penny rides on top of a piston as it undergoes vertical simple harmonic motion with an amplitude of 4.0 cm. If the frequency is low, the penny rides up and down without difficulty. If the frequency...
-
What are ownership-specific advantages, location specific advantages, and internalization advantages?
-
Time ( s ) Velocity ( m / s ) 1 2 3 4 5 6 7 8 Calculate the velocity
-
The table below gives the data about Etruria's balance of payments. (All figures are in billions of dollars.) Foreign investment in Etruria Secondary (transfers) income received from abroad Primary...
-
Olive Corporation buys a material for P20 per unit. Sixteen thousand parts a year are needed. Carrying costs is P3.00 per unit and the ordering cost is P15. Required: Compute the economic order...
-
As a healthcare leader or manager, most of us are charged with supervising employees. The literature suggests the importance of hiring and retaining employees with high levels of emotional...
-
7-8. Evaluate the sum exactly. (10 points each) 7. 18 (1) n (33) "
-
Calculate the indicated areas. All data are accurate to at least two significant digits. The widths of a kidney-shaped swimming pool were measured at 2.0-m intervals, as shown in Fig. 2.107....
-
The roof of a refrigerated truck compartment is of composite construction, consisting of a layer of foamed urethane insulation (t2 = 50 mm, ki = 0.026 W/m K sandwiched between aluminum alloy panels...
-
Complete the calculation of the mass of Earth as outlined in Section 2.7.
-
The Kingda Ka roller coaster in New Jersey is the worlds tallest ride of its kind. As the passenger cars are launched from rest at the start, they are accelerated uniformly to a speed of 57 m/s (128...
-
During our discussion of the motion of a falling body near Earths surface, we said that the gravitational force acting on itits weightis constant. But the law of universal gravitation tells us that...
-
Practice Problem 1 The stockholders equity accounts of Bramble Corp. on January 1, 2017, were as follows. Preferred Stock (6%, $100 par noncumulative, 4,400 shares authorized) $264,000 Common Stock...
-
JVCU Which of the following is considered cash for financial reporting purposes? 1 JVCU Which of the following is considered cash for financial reporting purposes? 1
-
Required information The Foundational 15 [LO8-2, LO8-3, LO8-4, LO8-5, LO8-7, LO8-9, L08-10) (The following information applies to the questions displayed below.) Morganton Company makes one product...

Study smarter with the SolutionInn App


