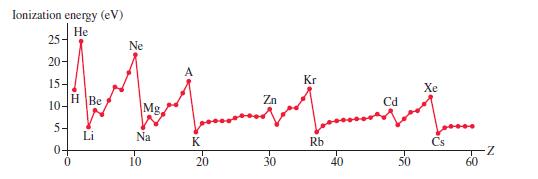
Figure 41.22 shows that the ionization energy of cadmium (Z = 48) is larger than that of
Question:
Figure 41.22 shows that the ionization energy of cadmium (Z = 48) is larger than that of its neighbors. Why is this?
Figure 41.22:
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

Physics for Scientists and Engineers A Strategic Approach with Modern Physics
ISBN: 978-0133942651
4th edition
Authors: Randall D. Knight
Question Posted:




