Question: FIGURE P16.58 shows two masses hanging from a steel wire. The mass of the wire is 60.0 g. A wave pulse travels along the wire
FIGURE P16.58 shows two masses hanging from a steel wire.
The mass of the wire is 60.0 g. A wave pulse travels along the wire from point 1 to point 2 in 24.0 ms. What is mass m?
The mass of the wire is 60.0 g. A wave pulse travels along the wire from point 1 to point 2 in 24.0 ms. What is mass m?
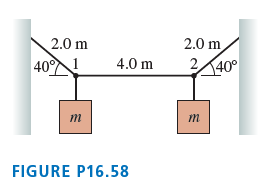
2.0 m 140 2.0 m 4.0 m 40 1 2, FIGURE P16.58
Step by Step Solution
★★★★★
3.46 Rating (162 Votes )
There are 3 Steps involved in it
1 Expert Approved Answer
Step: 1 Unlock

Model The wave pulse is a traveling wave on a stretch... View full answer

Question Has Been Solved by an Expert!
Get step-by-step solutions from verified subject matter experts
Step: 2 Unlock
Step: 3 Unlock
Document Format (2 attachments)
1442_6054778b8d501_693248.pdf
180 KBs PDF File
1442_6054778b8d501_693248.docx
120 KBs Word File


