Question: FIGURE P18.61 shows two different processes by which 1.0 g of nitrogen gas moves from state 1 to state 2. The temperature of state 1
FIGURE P18.61 shows two different processes by which 1.0 g of nitrogen gas moves from state 1 to state 2. The temperature of state 1 is 25?C. What are
(a) Pressure p1
(b) Temperatures (in ?C) T2, T3, and T4?
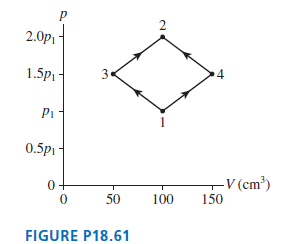
2.0p 1.5p 3. Pi 0.5p1 - V (cm) 50 100 150 FIGURE P18.61 2.
Step by Step Solution
3.34 Rating (163 Votes )
There are 3 Steps involved in it
Model Assume the nitrogen gas is an ideal gas Solve a ... View full answer

Get step-by-step solutions from verified subject matter experts


