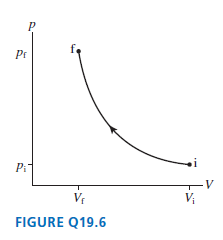
Figure Q19.6 shows an adiabatic process. a. Is the final temperature higher than, lower than, or equal
Question:
a. Is the final temperature higher than, lower than, or equal to the initial temperature?
b. Is any heat energy added to or removed from the system in this process? Explain.
Transcribed Image Text:
Pr Pi Vr V; FIGURE Q19.6
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 83% (6 reviews)
a Lower b No The tempe...View the full answer

Answered By

John Kimutai
I seek to use my competencies gained through on the job experience and skills learned in training to carry out tasks to the satisfaction of users. I have a keen interest in always delivering excellent work
4.70+
11+ Reviews
24+ Question Solved
Related Book For 

Physics for Scientists and Engineers A Strategic Approach with Modern Physics
ISBN: 978-0133942651
4th edition
Authors: Randall D. Knight
Question Posted:
Students also viewed these Physics questions
-
Five moles of an ideal monatomic gas with an initial temperature of 127C expand and, in the process, absorb 1200 J of heat and do 2100 J of work. What is the final temperature of the gas?
-
In an experiment to simulate conditions inside an automobile engine, 0.185 mol of air at a temperature of 780 K and a pressure of 3.00 X 106 Pa is contained in a cylinder of volume 40.0 cm3. Then 645...
-
A mass of 400(lbm) of 35-wl-% aqueous NaOH solution at 130(F) is mixed with 175(lbm) of 10-wt-% solution at 200(F). (a) What is the heat effect if the final temperature is 80(F)? (b) If the mixing is...
-
Featherstone Inc. reported the following data: Net income ................................... $296,000 Depreciation expense ................... 113,100 Gain on disposal of equipment ...... 58,500...
-
(a) A rubidium atom (m = 85u) is at rest with one electron in an excited energy level. When the electron jumps to the ground state, the atom emits a photon of wavelength = 780 nm. Determine the...
-
Sky's the Limit produces a variety of niche products for specific clients. During August, Sky's the Limit worked on Job X-10 for one of its regular customers. The following information is related to...
-
Does the run of the macroeconomy make a difference as to monetary policys potency? Explain.
-
The following section is taken from Koster Corp.s balance sheet at December 31, 2011. Current liabilities Interest payable.. $ 72,000 Long-term liabilities Bonds payable, 9%, due January 1,...
-
Describe simple present value, future value, annuities, perpetuities, and mortgages, and give an example of how to solve each type of
-
1. What types of conflict is the team experiencing in the challenge video? 2. What aspects of the negotiation process would best resolve the conflict and why would this work? 3. What conflict...
-
You throw a baseball straight up in the air so that it rises to a maximum height much greater than your height. Is the magnitude of the balls acceleration greater while it is being thrown or after it...
-
Figure Q19.7 shows two different processes taking an ideal gas from state i to state f. Is the work done on the gas in process A greater than, less than, or equal to the work done in process B?...
-
In Exercises find the derivative of the function. (t) = t 2/3 - t 1/3 + 4
-
Describe how the ideas within this Preamble align with your own personal values and career goals as a social work professional.
-
Maersk managers are adapting with various types of industry and market changes to nurture a contemporary approach to management . Analyze the various contemporary management practices of Maersk.
-
then P Let p 2 be an integer such that for any a, b integers, if divides a or p divides b. Show that p is prime. P divides ab
-
For Q2, you will use logistic regression to segment customers into two classes. This question is adapted from a Kaggle contest to evaluate current customers for an auto dealership that is opening a...
-
Figure how to fill out the rest Cash Budget 2018 Cash Balance, Beginning $ Q1 50,000 Q2 Q3 Q4 Cash Collections Cash Available 2,599,218 2,649,218 Manufacturing Outflows: Direct Materials 1,645,183...
-
Factor the given expressions completely. 144n 2 169p 4
-
Select the correct answer for each of the following questions. 1. On December 31, 20X3, Saxe Corporation was merged into Poe Corporation. In the business combination, Poe issued 200,000 shares of its...
-
A carpenter wishes to tighten the heavy head of his hammer onto its light handle. Which method shown in Figure Q4.7 will better tighten the head? Explain. FIGURE Q4.7
-
A ball sits near the front of a child's wagon. As she pulls on the wagon and it begins to move forward, the ball rolls toward the back of the wagon. Explain why the ball rolls in this direction.
-
Three arrows are shot horizontally. They have left the bow and are traveling parallel to the ground as shown in Figure Q4.6. Air resistance is negligible. Rank in order, from largest to smallest, the...
-
Be prepared to explain the texts comprehensive To illustrate the issues related to interest capitalization, assume that on November 1, 2016, Shalla Company contracted Pfeifer Construction Co. to...
-
On April 1, 2020. Indigo Company received a condemnation award of $473,000 cash as compensation for the forced sale of the company's land and building, which stood in the path of a new state highway....
-
The market price of a stock is $24.55 and it is expected to pay a dividend of $1.44 next year. The required rate of return is 11.23%. What is the expected growth rate of the dividend? Submit Answer...

Study smarter with the SolutionInn App


