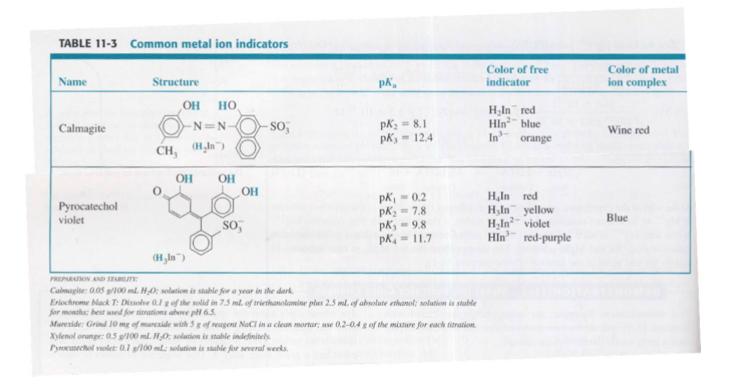
Question: Calcium ion was titrated with EDTA at pH 11, using Calmagite as indicator (Table 11-3). Which is the principal species of Calmagite at pH 11?
Calcium ion was titrated with EDTA at pH 11, using Calmagite as indicator (Table 11-3). Which is the principal species of Calmagite at pH 11? What color was observed before the equivalence point? After the equivalence point?
Step by Step Solution
3.33 Rating (168 Votes )
There are 3 Steps involved in it

Get step-by-step solutions from verified subject matter experts


